
Concept explainers
(a)
Interpretation:
The alcohol formed due to reaction of carbonyl with
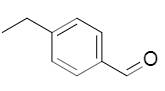
Concept introduction:
In the reduction of carbonyl compounds, hydrogen atoms are added to the carbonyl carbon, forming alcohols.
(b)
Interpretation:
The needed structural differences should be made to 4-methylcyclopentanone to derive the relevant product of reduction.
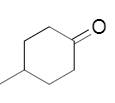
Concept introduction:
In the reduction of carbonyl compounds, Hydrogen atoms are added to the carbonyl carbon, forming alcohols finally. Ketones form secondary alcohols.
(c)
Interpretation:
The needed structural differences should be made to 5-methyl-3-hexanone to derive the relevant product of reduction.
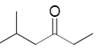
Concept introduction:
In the reduction of carbonyl compounds, ketones form secondary alcohols.
(d)
Interpretation:
The needed structural differences should be made to Octanal to derive the relevant product of reduction.
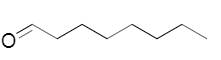
Concept introduction:
In the reduction of carbonyl compounds, an aldehyde is converted to primary alcohol.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
General, Organic, and Biological Chemistry - 4th edition
- • Whał arc the IUPAC namar of the ff. Carboxylic acias? a. COOH COOH CH3 b. I f. .COOH CHJCH2 ÇHCHCOOH CH3 NO2 tON C. COOH ноос d. COOHarrow_forwardWhich alkyl halide has the highest boiling point? A. CH3BrB. CH3FC. CH3ClD. CH3larrow_forward2. Complete the following reactions for the preparation of alcohols. Draw the structure of the product. Name the reactant and the product. a) CHy CH=CH-CHy + HyO Hydration of alkenes b) Cat. CHy-CHy + H2 drogenation carbonyl pups d)arrow_forward
- A Primary alcohol will get converted into what functional group in the following reaction? 1. DMSO, (COCI)2 Primary Alcohol ???? 2. EtgN carboxylic acid ketone aldehyde ester D. A Darrow_forwardWhich is propyl propanoate? A. CH₂CH₂CH₂OOCCH₂CH; B. CH₂CH₂CH₂COOCH₂CH₂ C. CH₂CH₂CH₂COCH₂CH₂ D. CHỊCH,CH,OCH,CHỊCH, A B C Darrow_forwardWhich of the following is not a physical property of alcohols or phenols? Select one: A. The solubilities of primary alcohols in water decrease with increasing molecular weight. B. Phenols are generally only slightly soluble in water. C. Due to hydrogen bonding, boiling points of alcohols are much higher than those of corresponding alkanes. D. The hydroxyl group of an alcohol is nonpolar.arrow_forward
- Give the IMPAC name for each compound. 1. COOH COOH 2. Br 3. CH3 CHz COOHarrow_forwardIf the following structure undergoes oxidation reaction, which of the product(s) will form: ОН H2C–CH2-CH2 CH3 O A. Aldehydes only O B. This molecule cannot go through oxidation reaction. OC. Ketone O D. Aldehyde & Carboxylic acid O E. Ether and waterarrow_forwardWhich of the following is not a physical property of alcohols or phenols? A. The solubilities of primary alcohols in water decrease with increasing molecular weight. B. Due to hydrogen bonding, boiling points of alcohols is much higher than those of corresponding alkanes. C. The hydroxyl group of an alcohol is nonpolar. D. Phenols are generally only slightly soluble in water.arrow_forward
- Classify each alcohol as to 1º, 2º, or 3º 1. 2. CH3CH2CH(CH3)CH2OHCH3 3. CH2CH2CHOHCH3arrow_forward3. Complete the following intramolecular dehydration reactions for alcohols. Draw the structure of the product. Name the reactant and the product. a) Cy-C-CH-oH 18UC b) CHy CH-Cy-CHy 180C OH c) 180c 4. Complete the following intermolecular dehydration reactions for alcohols. Draw the structure of the product. Name the reactant and the product. a) CHy-CH-OH b) Ho SわふわSわ 5. Complete the following oxidation reactions for alcohols. Draw the structure of the product. Name the reactant and identify the type of compound formed in the product. a) hparrow_forwardGive the IUPAC name for each ketone. C-CHCH,CH3 b. a.arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning




