
General, Organic, and Biological Chemistry - 4th edition
4th Edition
ISBN: 9781259883989
Author: by Janice Smith
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16, Problem 74P
Answer each question about phenylacetaldehyde, depicted in the ball-and-stick model, and Page 590 one of the components responsible for the fragrance of the plumeria flower.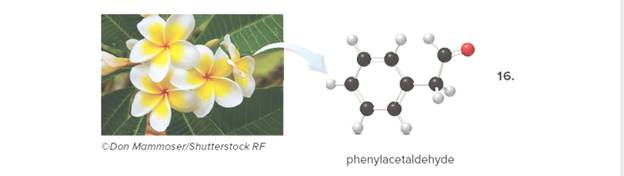
a. Draw a skeletal structure for phenylacetaldehyde.
b. How many trigonal planar carbons are present in phenylacetaldehyde?
c. What product is formed when phenylacetaldehyde is treated with two equivalents of
d. What product is formed when phenylacetaldehyde is treated with one equivalent of hydrogen gas in the presence of a metal catalyst?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
1. What is the role of the acetic acid in the oxidation of Cyclohexanol to Cyclohexanone? Write the balanced chemical reaction between acetic acid and sodium hypochlorite.2. How do you neutralize the acetic acid regenerated in the reaction? Write the balanced chemical reaction.
6. Draw the correct structures for the following:
a. what is the correct structure of 3-methyl 1-pentyne?
b. what is the correct structure of butyl methyl amine?
c. what is the correct structure of 3-methyl 1-pentyne?
d. what is the correct structure of 2-methyl 2-butanol?
e. what is the correct structure of pentanoic acid?
1. Write the skeletal structures of propanal, acetone and cyclohexanone. What is the major intermolecular force (IMF) found in them? Based on their major intermolecular force and molecular weight, what can you predict on their solubility in water?
Chemical Name
Skeletal Structures
Major IMF
Solubility in water
Propanal
Acetone
Cyclohexanone
2. What is the purpose of Tollens’ test (Part B)? What is the evidence of a positive result?
3. What is the purpose of oxidation test (Part C)? What is the evidence of a positive result?
Chapter 16 Solutions
General, Organic, and Biological Chemistry - 4th edition
Ch. 16.1 - Prob. 16.1PCh. 16.1 - Draw the structure of the three constitutional...Ch. 16.2 - Give the IUPAC name for each aldehyde. a. (...Ch. 16.2 - Give the structure corresponding to each TUPAC...Ch. 16.2 - Prob. 16.4PCh. 16.2 - Prob. 16.2PPCh. 16.2 - Give the structure corresponding to each name. a....Ch. 16.3 - Which compound in each pair has the higher boiling...Ch. 16.3 - Acetone and progesterone are two ketones that...Ch. 16.4 - Prob. 16.8P
Ch. 16.5 - What product is formed when each carbonyl compound...Ch. 16.5 - Prob. 16.4PPCh. 16.5 - Prob. 16.9PCh. 16.6 - Prob. 16.5PPCh. 16.6 - Prob. 16.10PCh. 16.6 - Prob. 16.11PCh. 16.7 - Prob. 16.12PCh. 16.8 - Prob. 16.6PPCh. 16.8 - Prob. 16.7PPCh. 16.8 - Prob. 16.13PCh. 16.8 - Prob. 16.8PPCh. 16.8 - Label the three acetalsin solanine, the toxic...Ch. 16.8 - Prob. 16.14PCh. 16.8 - Prob. 16.10PPCh. 16 - Prob. 15PCh. 16 - Prob. 16PCh. 16 - Prob. 17PCh. 16 - Prob. 18PCh. 16 - Prob. 19PCh. 16 - Prob. 20PCh. 16 - Prob. 21PCh. 16 - Prob. 22PCh. 16 - Prob. 23PCh. 16 - Prob. 24PCh. 16 - Give an acceptable name for each ketone. a. b. c....Ch. 16 - Prob. 26PCh. 16 - Prob. 27PCh. 16 - Draw the structure corresponding to each name. a....Ch. 16 - Prob. 29PCh. 16 - Prob. 30PCh. 16 - Prob. 31PCh. 16 - Prob. 32PCh. 16 - Draw out the structure of benzaldehyde, including...Ch. 16 - Prob. 34PCh. 16 - Which compound in each pair has the higher boiling...Ch. 16 - Prob. 36PCh. 16 - Prob. 37PCh. 16 - Prob. 38PCh. 16 - Prob. 39PCh. 16 - Prob. 40PCh. 16 - Prob. 41PCh. 16 - Prob. 42PCh. 16 - Prob. 43PCh. 16 - Prob. 44PCh. 16 - Prob. 45PCh. 16 - Consider the following ball-and-stick model....Ch. 16 - Prob. 47PCh. 16 - Prob. 48PCh. 16 - Prob. 49PCh. 16 - Prob. 50PCh. 16 - Prob. 51PCh. 16 - Prob. 52PCh. 16 - Prob. 53PCh. 16 - Prob. 54PCh. 16 - Prob. 55PCh. 16 - Prob. 56PCh. 16 - Prob. 57PCh. 16 - Prob. 58PCh. 16 - Prob. 59PCh. 16 - Label the functional group(s) in each compound as...Ch. 16 - Prob. 61PCh. 16 - Prob. 62PCh. 16 - What acetal is formed when each aldehyde or ketone...Ch. 16 - What acetal is formed when each aldehyde or ketone...Ch. 16 - Prob. 65PCh. 16 - Prob. 66PCh. 16 - Prob. 67PCh. 16 - Prob. 68PCh. 16 - Prob. 69PCh. 16 - Prob. 70PCh. 16 - Prob. 71PCh. 16 - Prob. 72PCh. 16 - Prob. 73PCh. 16 - Answer each question about phenylacetaldehyde,...Ch. 16 - Prob. 75PCh. 16 - Prob. 76PCh. 16 - Prob. 77PCh. 16 - Prob. 78PCh. 16 - Three constitutional isomers of molecular formula...Ch. 16 - Identify A—C in the following reaction sequenceCh. 16 - Androsterone is a male sex hormone that controls...Ch. 16 - Prob. 82PCh. 16 - Prob. 83PCh. 16 - Prob. 84PCh. 16 - Prob. 85PCh. 16 - Paraldehyde, a hypnotic and sedative once commonly...Ch. 16 - Prob. 87PCh. 16 - Prob. 88PCh. 16 - Prob. 89CPCh. 16 - Prob. 90CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 5. What products would result from the following processes? Write an equation for each reaction. a. 2-Methyl-2-butanol is subjected to controlled oxidation. b. 1-Propanol is heated to 140°C in the presence of sulfuric acid. c. 3-Pentanol is subjected to controlled oxidation. d. 3-Pentanol is heated to 180°C in the presence of sulfuric acid. e. 1-Hexanol is subjected to an excess of oxidizing agentarrow_forwardFLAVORANTS: Identify the aldehyde and ketone-containing flavoring compound from each following sources. Choose your best answer from the choices below A. 2-Octanone B. Citral C. Benzaldehyde D. Vanillin E. Cinnamaldehyde F. a-Damascone 1. Вerry 2. Mushroom 3. Lemongrass 4. Cinnamon 5. Almonds Idchyde or ketone formed froarrow_forward1. What are the substances present in the label of formaldehyde (formalin)? 2. What is the percentage composition of acetone in the nail polish remover? 3. According to your interview, what is/are the uses of formalin and acetone? A. Uses of Formalin- B. Uses of Acetone - 4. What type of bond is present in the two structures? 5. Why do you think aldehydes and ketones belong to the group of carbonyl containing compounds?arrow_forward
- Which of the following is not a physical property of alcohols or phenols? Select one: A. The solubilities of primary alcohols in water decrease with increasing molecular weight. B. Phenols are generally only slightly soluble in water. C. Due to hydrogen bonding, boiling points of alcohols are much higher than those of corresponding alkanes. D. The hydroxyl group of an alcohol is nonpolar.arrow_forward50. If this molecule was heated, which carboxyl group will be readily lost as carbon dioxid. C HO2C CO2H CO2H HO2C B a. b. B C. C d.arrow_forward9. Label the following reactions as: A. ionization of a carboxylic acid in water B. neutralization of a carboxylic acid with a base C. formation of an ester D. acid-catalyzed hydrolysis of an ester E. ionization of an amine in water F. neutralization of an amine with an acid G. formation of an amide H. hydrolysis of an amide CH3NH₂ + HCI CH3NH3 Cl CH3CH₂CNHCH 3 HOH CH₂COH CH3COH+ NaOH →→→→ CHICOH heat CHICOH CH3COH + CH3NH₂ CH₂COH CH3CH₂COCH₂CH₂CH + HOHH CH3CH₂COH + HOCH₂CH₂CH3 CHO + H₂O - CH3CO heat CHO CH₂CO *Na + HOH CH3NH₂ + H₂0 CH3NH3* + OH- + H3O+ CH3CH₂C + NH2CH3 R-H R-O-H CH3CNHCH3 + HOH R-N- I O CH₂COH CH3COH + HOCH3 CH3COCH3 + HOH сно сосни, O || R-C-R O 11 R-C-O-R Hydrocarbons Alcohols Amines Ketones Esters R-X R-O-R 0=0|0=0|0=0 R-C-H R-C-0-H R-C-N- I Alkyl halides Ethers Aldehydes Carboxylic Acids Amidesarrow_forward
- C. Draw a structural formula for the principal organic product formed when each compound is treated with K2CrzOr/H2SO4 if there is no reaction, say so. 1. butanal 2. benzaldehyde 3. cyclohexanone 4. cyclohexanol 5. 2-pentanonearrow_forwardIf the following structure undergoes oxidation reaction, which of the product(s) will form: ОН H2C–CH2-CH2 CH3 O A. Aldehydes only O B. This molecule cannot go through oxidation reaction. OC. Ketone O D. Aldehyde & Carboxylic acid O E. Ether and waterarrow_forwardWrite the complete chemical reaction and correct chemical formula and structure of the following: 1. Autooxidation of benzaldehyde 2. reaction between acetic acid and ethanol 3. reaction between aspirin and sodium hydroxide 4. reaction of acetone with methanolarrow_forward
- • Whał arc the IUPAC namar of the ff. Carboxylic acias? a. COOH COOH CH3 b. I f. .COOH CHJCH2 ÇHCHCOOH CH3 NO2 tON C. COOH ноос d. COOHarrow_forwardWhich of the following is not a physical property of alcohols or phenols? A. The solubilities of primary alcohols in water decrease with increasing molecular weight. B. Due to hydrogen bonding, boiling points of alcohols is much higher than those of corresponding alkanes. C. The hydroxyl group of an alcohol is nonpolar. D. Phenols are generally only slightly soluble in water.arrow_forwardWhat is the systematic IUPAC name for the given compound? CH3 CH3 CH;CHCH,CH,CH2 -Ń-CH3 a. N-methyl-4-methylhexan-1-amine b. 2,4-dimethylhexan-1-amine c. 2,2,N-trimethylpentan-1-amine d. N,N,4-trimethylpentan-1-aminearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
CBSE Class 12 Chemistry || Polymers || Full Chapter || By Shiksha House; Author: Best for NEET;https://www.youtube.com/watch?v=OxdJlS0xZ0Y;License: Standard YouTube License, CC-BY