
Concept explainers
(a)
Interpretation:
If the given compound is acetal, or ether should be determined.
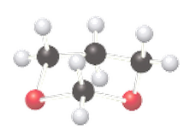
Concept Introduction:
The combination of two
(b)
Interpretation:
If the given compound is acetal, or ether should be determined.
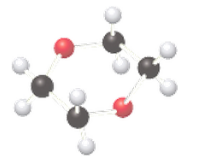
Concept Introduction:
The combination of two functional groups results in the formation of a hemiacetal. In a hemiacetal, a carbon atom is bonded to an alcohol group and an ether group. It is derived from the aldehyde. When two ether and one alcohol group is bonded to a carbon atom, then it results in the formation of acetal. It is derived from hemiacetal.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
General, Organic, and Biological Chemistry - 4th edition
- An acid catalyst in nucleophilic addition of aldehydes and ketones is used for: Select one: a. Protonation of carbonyl carbon b. Making the aldehyde and ketone more susceptible to nucleophiles c. Increasing the nucleophilicity of the nucleophile d. To provide a medium for the reaction.arrow_forwardLabel the functional group(s) in each compound as ethers or acetals.arrow_forwardGive the IUPAC name for each sulde.arrow_forward
- Explain why the ether with formula C2H6O is very slightly soluble in water, whereas the alcohol with the same formula is infinitely soluble in water.arrow_forwardLabel each of the following structures as a cyclic hemiacetal, hemiketal, acetal, ketal, or none of these: a. b. c.arrow_forwardare acetals and ketals stable to acid or base? Why?arrow_forward
- 1. Differentiate Ethyl alcohol from methyl alcohol. 2. Compare aldehydes and ketones as to (Use acetaldehyde and acetone as examples). A. Reaction with cone. NaOH, heated B. Reaction with Tollen’s reagentarrow_forwardDraw the structure of a compound tting each description:a. an aldehyde with molecular formula C4H8Ob. a ketone with molecular formula C4H8Oc. a carboxylic acid with molecular formula C4H8O2d. an ester with molecular formula C4H8O2arrow_forwardGive the IUPAC and common name for the following compounds. a. HO, b. но OHarrow_forward
- 10.38 Give the IUPAC name for each compound. a. b. C. d. e. OH f. OHarrow_forward1. Simple ketones, like acetone, are often used as industrial solvents for many organically based products such as adhesives and paints. They are considered "universal solvents," because they dissolve so many diverse materials. Explain why these chemicals are good solvents.arrow_forwardExplain the General Features of Reactions of Ethers ?arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning


