
Concept explainers
Show how you would modify the synthesis given in the previous problem to synthesize the following drugs:
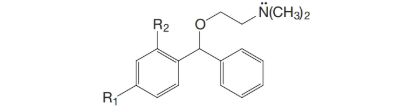
(a) Bromodiphenhydramine (an antihistamine), where
(b) Orphenadrine (an antispasmodic, used in controlling Parkinson’s disease), where
Want to see the full answer?
Check out a sample textbook solution
Chapter SRP Solutions
Organic Chemistry
Additional Science Textbook Solutions
Fundamentals of Heat and Mass Transfer
Organic Chemistry As a Second Language: Second Semester Topics
Chemistry & Chemical Reactivity
Chemistry: A Molecular Approach
Basic Chemistry (5th Edition)
- Nonconjugated , -unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated , -unsaturated isomers. Propose a mechanism for this isomerization.arrow_forwardAldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield acetals. Predict the product of the following reaction, and propose a mechanism:arrow_forwardSynthesize the following compound from benzonitrile (C6H5CN):arrow_forward
- Following is the structural formula of the principal sex pheromone of the Douglas fir tussock moth (Orgyia pseudotsugata), a severe defoliant of the fir trees of western North America. (Z)-6-Heneicosene-11-one Several syntheses of this compound have been reported, starting materials for three of which are given here. (a) `H + OH Br (b) HO Br (c) Br + Show a series of steps by which each set of starting materials could be converted into the target molecule.arrow_forwardWhen A is reacted with hot aqueous NaOH, a compound B of molecular formula C8H11NO is produced. With this information, write the correct structure of B and propose the reaction mechanism (step by step, with the correct use of arrows) to obtain B.arrow_forwardPredict the products of the following acid-base reactions. If the equilibrium would not result in the formation of appreciable amounts of products, you should so indicate. In each case label the stronger acid, the stronger base, the weaker acid, and the weaker base: (a) CH3CH=CH2 + NANH2 (d) CH3C=C: + CH;CH2OH → (e) CH3C=C:- + NH¾CI – | (b) CH;C=CH + NaNH2 (c) CH3CH2CH3 + NANH2 → | HASarrow_forward
- (c) The structures of lycopene, beta-carotene and retinol are shown below. Lycopene Beta-carotene H3C CH; CH3 CH3 OH Retinol (vitamin A) CH3 Using appropriate reaction mechanisms: (i) Show how lycopene is converted into beta-carotene. (ii) Briefly explain how retinol is formed from beta-carotene.arrow_forwardGive the expected major product of the following reaction: OH 1) LIAIH,arrow_forwardPropose a mechanism for the reaction of(a) 1-methylcyclohexanol with HBr to form 1-bromo-1-methylcyclohexane.(b) 2-cyclohexylethanol with HBr to form 1-bromo-2-cyclohexylethane.arrow_forward
- 3. Predict the products of the following reactions. When more than one product is expected, predict which will be the major product? ОН H,SO4 H;PO4 (a) (b) heat OH heat CH3 Br CH3 Br NaOCH3 NaOC(CH3)3 (c) (d)arrow_forwardThe tosylate of a primary alcohol normally undergoes an S2 reaction with hydroxide ion to give a primary alcohol. Reaction of this tosylate, however, gives a compound of molecular formula C,H,0. OH NaOH C,H190 OTs Propose a structural formula for this compound and a mechanism for its formation.arrow_forwardDraw the structure of (2Z, 6E)-3-ethyl-7-methyldeca-2, 6-dien-1-ol, the sex pheromone of the codling moth, a common agricultural pest that destroys apple crops. By spraying an apple orchard with this pheromone, the mating of male and female moths is disrupted, and moth populations can be controlled.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

