
Concept explainers
Compound Y
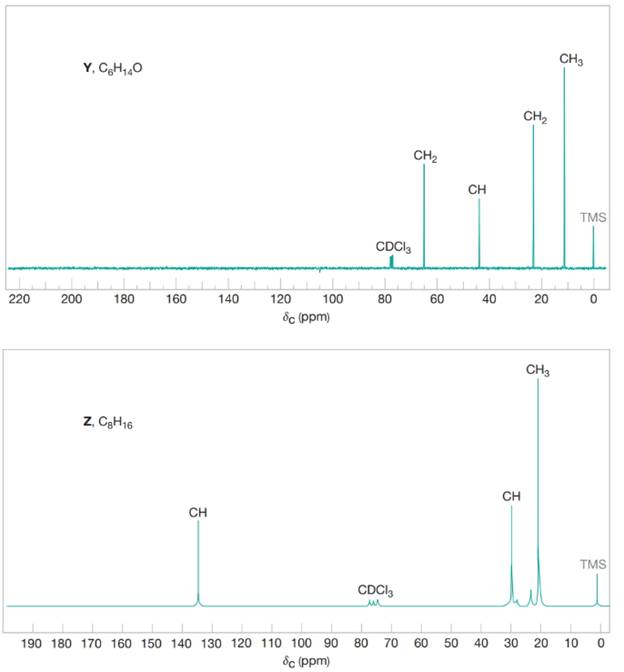
Figure 1 The broadband proton-decoupled
Want to see the full answer?
Check out a sample textbook solution
Chapter SRP Solutions
Organic Chemistry
Additional Science Textbook Solutions
Chemistry & Chemical Reactivity
Chemistry: A Molecular Approach
Chemistry
Organic Chemistry (9th Edition)
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
Introductory Chemistry (6th Edition)
- 3-Chlorocyclopropene, on treatment with AgBF4, gives a precipitate of AgCl and a stable solution of a product that shows a single 1H NMR absorption at 11.04 δ. What is a likely structure for the products, and what is its relation to HĂ¼ckel’s rule?arrow_forwardAddition of m-xylene to the strongly acidic solvent HF/SbF5 at 45C gives a new species, which shows 1H-NMR resonances at 2.88 (3H), 3.00 (3H), 4.67 (2H), 7.93 (1H), 7.83 (1H), and 8.68 (1H). Assign a structure to the species giving this spectrum.arrow_forwardCompound L has the formula C₁1H₁5 NO. The ¹H NMR, 13C NMR and the IR spectra for Compound L are shown below. Propose a structure for Compound L. 12 4800 11 HSP-01-587 DEPT-90 DEPT-135 1 : T T 10 9 200 2 2 CDS-06-337 8 7 180 160 6 : 140 5 ppm 120 4 4 3 100 : 6 ppm 2 T T 1 to maineriberent waiho mlenmol wons havus saonic 80 D 0 -1 T 60 40 20 4 T 0arrow_forward
- A compound has a molecular formula of C8H8O The following spectra (1H NMR, Carbon 13 NMR, IR was collected. Give a possible structure. 13C NMR – 8 peaksarrow_forwardPART 2: Compounds A, B, and C are isomers with the formula C5H11Br. Their broadband proton-decoupled 13C NMR spectra are given below. Information from the DEPT 13C NMR spectra is given near each peak. Give structure for C.arrow_forwardAn unknown compound with molecular formula C9H180 forms a phenylhydrazone, but gives a negative with Tollens' test. The IR spectrum has a strong band near 1710 cm. The broadband proton- decoupled "C NMR spectrum is given below. Propose a structure for the unknown compound. Explain your answer based on the given spectroscopic data CH3 A, C3H180 CH2 CH CDCI3 TMS 220 200 180 160 140 120 100 80 60 40 20 S, (ppm)arrow_forward
- 3. Propose a structure for Compound X which has a molecular formula C1oH12O with an IR absorption at 1687 cm-1. The 'H NMR spectrum of O is given below. 'HNMR of Compound X 3H зн 2H 2H 5H 8 7 6 5 3 2 ppmarrow_forwardOn the basis of the molecular formula, 1H NMR data, and IR data provided, propose a consistent structure. Moleculal Formula: C2H3Cl31H NMR data: δ 3.95 (d, 2H), 5.77 (t, 1H)IR data: 2950 cm–1, and several peaks below 820 cm–1.arrow_forwardA hydrocarbon, compound B, has molecular formula C6H6, and gave an NMR spectrum with two signals: delta 6.55 pm and delta 3.84 pm with peak ratio of 2:1. When warmed in pyridine for three hr, compound B quantitatively converts to benzene. Mild hydrogenation of B yielded another compound C with mass spectrum of m/z 82. Infrared spectrum showed no double bonds; NMR spectrum showed one broad peak at delta 2.34 ppm. With this information, address the following questions. a) How many rings are in compound C? b) How many rings are probably in B? How many double bonds are in B? c) Can you suggest a structure for compounds B and C? d) In the NMR spectrum of B, the up-field signal was a quintet, and the down field signal was a triplet. How must you account for these splitting patterns?arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole


