
Organic Chemistry
12th Edition
ISBN: 9781118875766
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter SRP, Problem 31P
A compound (N) with the molecular formula
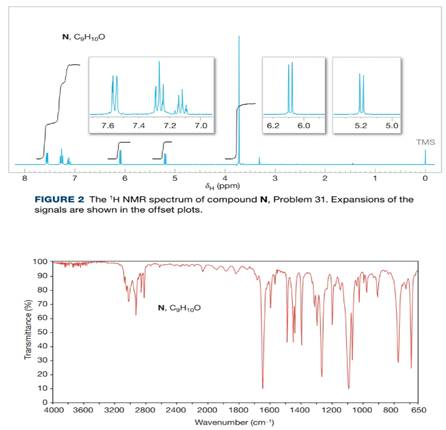
Figure 3 The IR spectrum of compound N, Problem 31.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Name the following compounds A and B.
How could you distinguish these two molecules by using 1H NMR and IR techniques?
Propose an analytical technique to determine the iron content of these compounds.
Calculate the mass percentages of C and H of compound B (C: 12.01 g/mol; H: 1.008 g/mol; Fe: 55.845 g/mol).
A compound A of mass 114 has the following elemental analysis: C = 63.14%, H=8.83%.
The IR, 1H NMR and 13C NMR spectra are given below.
1) Assign and interpret all the data from the 1H NMR and 13C NMR spectra. Propose a formula
for A.
7.0
175
с
3000
1,3; 13
I=1
6.0
150
I=1
с
5.0
CH₂
125
2000
Explain why the answer is this one :
CH,—CH,—0
4,2; 60
mavermy
I=2
cm-1
4.0
100
1500
3.0
75
135
167 |
CH₂
6
125
CH 3
1,95 ; 18
I=3
1000
2.0
50
H
5,5
I=3
1.0
CH₂
CH₂
25
500
Ppm
ppm
The IR, 'H NMR and 1C NMR spectra of compound P are shown below. The
molecular formula for compound Pis CHNO2
a.
Identify three (3) important absorption peaks on the IR spectrum and indicate
the bond and wavenumber associated with each peak.
Elucidate the structure of compound P and assign esch peak in the 'H and
1C NMR spectra. (Hint: The compound consists of more than one functional
group).
b.
T747cm
2000
a
quartet
triplet
CDCI,
Chapter SRP Solutions
Organic Chemistry
Ch. SRP - 1. Arrange the compounds of each of the following...Ch. SRP - 2. Arrange the compounds of each of the following...Ch. SRP - Predict the final product from each of the...Ch. SRP - Prob. 4PCh. SRP - Write detailed mechanisms for each of the...Ch. SRP - Prob. 6PCh. SRP - Prob. 7PCh. SRP - Prob. 8PCh. SRP - Prob. 9PCh. SRP - Give stereochemical structures for compounds AD:
Ch. SRP - Prob. 11PCh. SRP - The remaining steps in the industrial synthesis of...Ch. SRP - Prob. 13PCh. SRP - Prob. 14PCh. SRP - Prob. 15PCh. SRP - Prob. 16PCh. SRP - 17. Show how you would modify the synthesis given...Ch. SRP - Prob. 18PCh. SRP - Give structures for compounds AD. Compound D gives...Ch. SRP - The tranquilizing drug meprobamate (Equanil or...Ch. SRP - Prob. 21PCh. SRP - 22. Outlined here is the synthesis of a central...Ch. SRP - 23. What are compounds A and B? Compound B has a...Ch. SRP - Prob. 24PCh. SRP - 25. The Dow process for synthesizing phenol, which...Ch. SRP - Prob. 26PCh. SRP - Prob. 27PCh. SRP - 28. Compound Y shows prominent IR absorption...Ch. SRP - Prob. 29PCh. SRP - Consider this reaction involving peracetic acid:...Ch. SRP - 31. A compound (N) with the molecular formula...Ch. SRP - 32. Compound X is insoluble in aqueous sodium...Ch. SRP - Write the structures of the three products...Ch. SRP - Compound C (C9H11NO) gives a positive Tollens test...Ch. SRP - A compound X (C10H14O) dissolves in aqueous sodium...Ch. SRP - Compound Z (C5H10O) decolorizes bromine. The IR...Ch. SRP - 37. Compound W was isolated from a marine annelid...Ch. SRP - 38. Phenols generally are not changed on treatment...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Write electron configurations for each element. Use the symbol of the previous noble gas in brackets to represe...
Introductory Chemistry (5th Edition) (Standalone Book)
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
For Practice 1.1
Is each change physical or chemical? Which kind of property (chemical or physical) is demonst...
Principles of Chemistry: A Molecular Approach (3rd Edition)
A solution containing 100 lbm KNO3/100 Ibm H2O at 80°C is fed to a cooling crystallizer operated at 25°C. Slurr...
Elementary Principles of Chemical Processes, Binder Ready Version
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Compound I (C11H14O2) is insoluble in water, aqueous acid, and aqueous NaHCO3, but dissolves readily in 10% Na2CO3 and 10% NaOH. When these alkaline solutions are acidified with 10% HCl, compound I is recovered unchanged. Given this information and its 1H-NMR spectrum, deduce the structure of compound I.arrow_forward3-Chlorocyclopropene, on treatment with AgBF4, gives a precipitate of AgCl and a stable solution of a product that shows a single 1H NMR absorption at 11.04 δ. What is a likely structure for the products, and what is its relation to HĂ¼ckel’s rule?arrow_forwardThe proton NMR spectrum for a compound with formula C10H12O2is shown below.The infrared spectrum has a strong band al 1711 cm-1. The normal carbon-13 NMRspectral results are tabulated along with the DEPT-135 and DEPT-90 information. Drawthe structure of this compound.arrow_forward
- 22. A compound with the molecular formula C8H8O produces an IR spectrum with signals at 3063, 1686, and 1646 cm-1. The 1H NMR spectrum of this compound exhibits a singlet at 2.6ppm(l=3H), and a multiplet at 7.5(l= 5H). Draw the structure and give the common name of this compound . Show the correlations between the structure and spectra.arrow_forwardMass spectrometry of an unknown compound revealed a molecular ion at m/z 121.09. The IR spectrum, the 13C NMR spectrum, and the 'H NMR spectrum are shown below. Draw the structure of this compound.arrow_forwardPropose a structure for D (molecular formula CgH,CIO,) consistent with the given spectroscopic data. 13C NMR signals at 30, 36, 128, 130, 133, 139, and 179 ppm 1Η ΝMR of D 4H 2H 2 H 1H 12 10 8 4 2 ppmarrow_forward
- Part 3A Set 1. Can IR spectroscopy be used to differentiate between the two compounds? Briefly explain why or why not. What significant absorptions would be observed in the IR spectrum?arrow_forward2 The IR and 1H-NMR spectra of a compound with molecular formula C4H7ClO2 are shown below. Your objective as a group is to propose a structure for this compound, explaining how you reach your decision. Using all the information you have been given, in a post with others in your group share your initial ideas about the possible structure of the compound. Then use comments to interact with the other students in the group and propose a final answer to the problem. In the comment phase, you should comment on the postings of at least two other students.arrow_forward28. Shown below is the NMR spectrum of a compound with formula C.H.,0 4.0 Spectrum in the infrared has intense absorption at 1740 cm 1 and several intense bands between 1333 and 1035 cm -1 . Draw the structure of the compound. note: point out the structures on the chart for better understandingarrow_forward
- a compound C9H10O2 has the IR, 13C NMR, and 1H NMR spectra below.What is the structure? Explain answer based on spectral assignments. Assign at least 2 bands on the IR and all protons in the H NMR spectrumarrow_forward35. Compounds A. and B. are closely related and have formulas of C₁0H₁2O and C₁1H140 respectively. Both show an IR absorption at 1710 cm-¹. Based on NMR of A. and B. provide structures for each a. - ¹H NMR t S 2 11 10 Integrals 9 T m, (overlapped resonances) 5 8 7 6 5 ppm 4 3 2 T -N 2 T 3 1 0arrow_forwardCompound X (molecular formula C10H120) was treated with NH2NH2, ¯OH to yield compound Y (molecular formula C10H14). Match the 1H NMR spectra of X and Y to the corresponding structures of X and Y. Compound NH2NH2 Compound 'H NMR of X 6 H OH Y 1 H 5H 8. 6. 4 ppm or H NMR of Y 6 H 2H 5H 1 H multiplet multiplet 8. 6. 4. 3. 1 nnm 2. 2. 3, O:arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole

NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY