
Concept explainers
(a)
Interpretation:
The structure for the given IUPAC name is to be drawn.
Concept introduction:
The root name in the IUPAC name is the parent
Answer to Problem A.44P
The structure for the given IUPAC name is as follows:
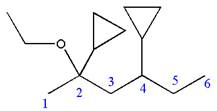
Explanation of Solution
The given IUPAC name is
The root name ‘hexane’ indicates the parent chain is of six carbon atoms.
Thus, the chain is numbered as shown below:
![]()
In the IUPAC name
Hence, the structure of
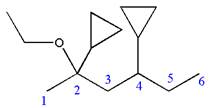
The structure for the given molecules is drawn from the root name, prefix and locator of substituent.
(b)
Interpretation:
The structure for the given IUPAC name is to be drawn.
Concept introduction:
The root name in the IUPAC name is the parent alkane. The longest continuous carbon chain must be drawn from the root name and numbered. The substituents are attached to the desired carbon number according to locator numbers given in the IUPAC name. The prefix indicates the number of identical substituents in the molecule.
Answer to Problem A.44P
The structure for
Explanation of Solution
The given IUPAC name is
The root name cyclohexane indicates the parent is a ring of six carbon atoms.
Hence, the structure of
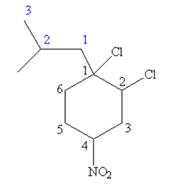
The structure for the given molecules is drawn from the root name, prefix and locator of substituent.
(c)
Interpretation:
The structure for the given IUPAC name is to be drawn.
Concept introduction:
The root name in the IUPAC name is the parent alkane. The longest continuous carbon chain must be drawn from the root name and numbered. The substituents are attached to the desired carbon number according to locator numbers given in the IUPAC name. The prefix indicates the number of identical substituents in the molecule.
Answer to Problem A.44P
The structure for the given IUPAC name is as follows:
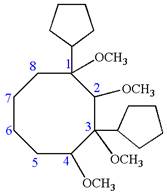
Explanation of Solution
The given IUPAC name is
The root name cyclooctane indicates the parent is a ring of eight carbon atoms.
Hence, the structure of
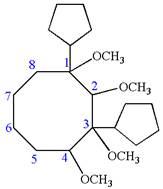
The structure for the given molecules is drawn from the root name, prefix and locator of substituent.
(d)
Interpretation:
The structure for the given IUPAC name is to be drawn.
Concept introduction:
The root name in the IUPAC name is the parent alkane. The longest continuous carbon chain must be drawn from the root name and numbered. The substituents are attached to the desired carbon number according to locator numbers given in the IUPAC name. The prefix indicates the number of identical substituents in the molecule.
Answer to Problem A.44P
The structure for the given IUPAC name is as follows:
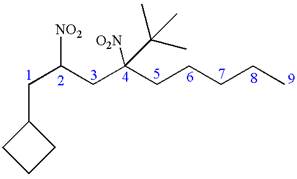
Explanation of Solution
The given IUPAC name is
The root name nonane indicates the parent is a chain of nine carbon atoms.
![]()
Hence, the structure of
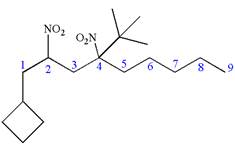
The structure for the given molecules is drawn from the root name, prefix and locator of substituent.
(e)
Interpretation:
The structure for the given IUPAC name is to be drawn.
Concept introduction:
The root name in the IUPAC name is the parent alkane. The longest continuous carbon chain must be drawn from the root name and numbered. The substituents are attached to the desired carbon number according to locator numbers given in the IUPAC name. The prefix indicates the number of identical substituents in the molecule.
Answer to Problem A.44P
The structure for the given IUPAC name is as follows:
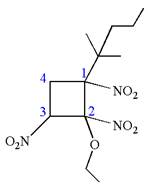
Explanation of Solution
The given IUPAC name is
The root name cyclobutane indicates the parent is a ring of four carbon atoms.
Hence, the structure of
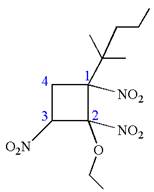
The structure for the given molecules is drawn from the root name, prefix and locator of substituents.
(f)
Interpretation:
The structure for the given IUPAC name is to be drawn.
Concept introduction:
The root name in the IUPAC name is the parent alkane. The longest continuous carbon chain must be drawn from the root name and numbered. The substituents are attached to the desired carbon number according to locator numbers given in the IUPAC name. The prefix indicates the number of identical substituents in the molecule.
Answer to Problem A.44P
The structure for the given IUPAC name is as follows:
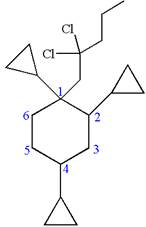
Explanation of Solution
The given IUPAC name is
The root name cyclohexane indicates the parent is a ring of six carbon atoms.
Hence, the structure of
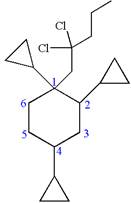
The structure for the given molecules is drawn from the root name, prefix and locator of substituent.
(g)
Interpretation:
The structure for the given IUPAC name is to be drawn.
Concept introduction:
The root name in the IUPAC name is the parent alkane. The longest continuous carbon chain must be drawn from the root name and numbered. The substituents are attached to the desired carbon number according to locator numbers given in the IUPAC name. The prefix indicates the number of identical substituents in the molecule.
Answer to Problem A.44P
The structure for the given IUPAC name is as follows:
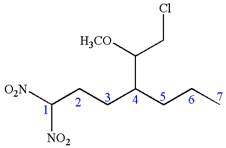
Explanation of Solution
The given IUPAC name is
The root name heptane indicates the parent is a chain of seven carbon atoms.
![]()
Hence, the structure of
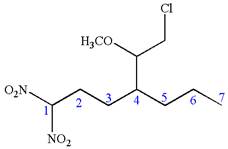
The structure for the given molecules is drawn from the root name, prefix and locator of substituent.
(h)
Interpretation:
The structure for the given IUPAC name is to be drawn.
Concept introduction:
The root name in the IUPAC name is the parent alkane. The longest continuous carbon chain must be drawn from the root name and numbered. The substituents are attached to the desired carbon number according to locator numbers given in the IUPAC name. The prefix indicates the number of identical substituents in the molecule.
Answer to Problem A.44P
The structure for the given IUPAC name is as follows:
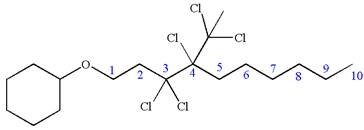
Explanation of Solution
The given IUPAC name is
The root name decane indicates the parent is a chain of ten carbon atoms.
![]()
Hence, the structure of
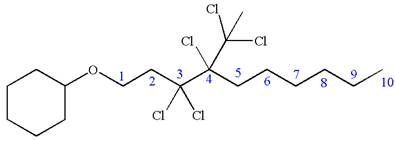
The structure for the given molecules is drawn from the root name, prefix and locator of substituent.
Want to see more full solutions like this?
Chapter A Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Predict which member of each group is most soluble in water, and explain the reasons for your predictions.(a) butan-1-ol, pentan-1-ol, or propan-2-ol(b) chlorocyclohexane, cyclohexanol, or cyclohexane-1,2-diol(c) phenol, cyclohexanol, or 4-methylcyclohexanolarrow_forwardDraw a structure for each compound (includes old and new names).(a) 3-methylpent-1-ene (b) cis-3-methyl-3-hexene (c) 3,4-dibromobut-1-ene(d) 1,3-cyclohexadienearrow_forwardDraw the structure that corresponds with each name.(a) 3-ethyloctane (b) 4-isopropyldecane (c) sec-butylcycloheptane(d) 2,3-dimethyl-4-propylnonane (e) 2,2,4,4-tetramethylhexane (f) trans-1,3-diethylcyclopentane(g) cis-1-ethyl-4-methylcyclohexane (h) isobutylcyclopentane (i) tert-butylcyclohexane(j) pentylcyclohexane (k) cyclobutylcyclohexane (l) cis-1-bromo-3-chlorocyclohexanearrow_forward
- The IUPAC name of the compound H- is (a) 5-formylhex-2-en-3-one (b) 5-methyl-4-oxohex-2-en-5-al (c) 3-keto-2-methylhex-5-enal (d) 3-keto-2-methylhex-4-enal ( The correct statement regarding electrophile is (a) electrophile is a negatively charged species and can form a bond by accepting a pair of electrons from another electrophile (b) electrophiles are generally neutral species and can form a bond by accepting a pair of electrons from a nucleophile (c) electrophile can be either neutral or ar positively charged species and can form a bond by accepting a pair of electrons from a nucleophile (d) electrophile is a negatively charged species and can form a bond by accepting a pair of electrons from a nucleophile.. Which among the given molecules can exhibit tautomerism? Ph Ph I II III (a) III only (c) Both I and II (b) Both I and III (d) Both II and III 1) Which of the following biphenyls is optically active?arrow_forwardDraw the structures of the following compounds. (a) 1-chloro-1 isopropylcyclopentanearrow_forwardc) For the IUPAC names below, provide an unambiguous structure! 1) (1R, 2R, 4S)-1,2-dibromo-4-methyl-cyclohexane 2) (2R, 3Z)-5-Fluoro-3-penten-1,2-diolarrow_forward
- is (b) 2, 3-dimetlyp (c) 2-ethyl-3-methylbutanal (d) 2-ethyl-3-methylbutan-3-al The IUPAC name of is CHO 2. Br (a) 2-methyl-3-bromohexanal (b) 3-bromo-2-methylbutanal (c) 2-methyl-3-bromobutanal (d) 3-bromo-2-methylpentanal 3. The IUPAC name of the compound HO is (a) 2-ethenyl-3-methyl cyclohexa-1, 3-diene (b) 2, 5-dimethyl hepta-2, 6-dienoic acid (c) 2, 6-dimethyl hepta-2, 5-dienoic acid (d) 2, 3-dimethyl epoxyethane 4. The IUPAC name of the compoundarrow_forwardGiven each of the IUPAC names provided, draw the corresponding structure. (a) 1-(1,1-dimethylethyl)-2,4-diethylcyclohexane; (b) 1,4-dibutyl-2-(1-methylpropyl)cyclooctane; (c) 1,1-dicyclopropyl-3-(1,1-dimethylethyl)cycloheptanearrow_forwardWrite the bond line formula of the following compounds: (a) 4-methyl-2-hexene, two geometrical (stereoisomers) isomers (b) 3-fluoro-2-methylheptanol (3-fluoro-2-methylheptan-1-ol) (c) 4-methyl-hex-1-yn-3-olarrow_forward
- Some of the following examples can show geometric isomerism, and some cannot. Forthe ones that can, draw all the geometric isomers, and assign complete names using theE-Z system.(a) 3-bromo-2-chloropent-2-ene (b) 3-ethylhexa-2,4-diene(c) 3-bromo-2-methylhex-3-ene (d) penta-1,3-diene(e) 3-ethyl-5-methyloct-3-ene (f) 3,7-dichloroocta-2,5-dienearrow_forwardWrite the reagent or draw structures of the starting material or organic product(s) in the following reactions. If more than one product is formed, identify the major product where possible. (a) (b) HO OH OH H2SO4 ? Cl₂ ? FeCl3arrow_forwardDraw the structural formula for each of the following compounds:(i) 3-methyl-5-(2,2-dimethylpropyl) nonane(ii) 1,2-dichloro-3-methylcyclohexanearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





