
Spheres A and B arc initially at 800 K, and they are simultaneously quenched in large constant temperature baths, each having a temperature of 320 K. The following parameters are associated with each of the spheres and their cooling processes.
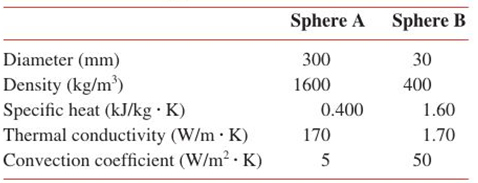
(a) Show in a qualitative manner, on
(b) Calculate the time required for the surface of each sphere to reach 415 K.
(c) Determine the energy that has been gained by each of the baths during the process of the spheres cooling to 415 K.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Introduction to Heat Transfer
- An underwater sonar that maps the ocean bathymetry is encapsulated in a sphere with a diameter of 85 mm. During operation, the sonar generates heat at a rate of 300W. What is the sonar surface temperature when it’s located in a water column where the temperature is 15o C and the water current is 1 m/sec? The sonar was pulled out of the water without turning it off, thus, it was still working. The air temperature was 15o C and the air speed was 3 m/sec. What was the sonar surface temperature? Was there any reason for concern?arrow_forwardVacuum furnaces are used in manufacturing for metal heating in brazing and sintering processes, as well as for heat treatment of steel alloys. The vacuum, and absence of air/oxygen, facilitates a low contamination process and prevents detrimental surface oxidisation. Heat is often provided in these furnaces by electrical heating elements, and their thermal design is critical to the effective operation of the furnace. In one such case, this element is modelled as a long, cylindrical electrically heated rod, 20 mm in diameter installed in a vacuum furnace as shown. The surface of the heating rod has an emissivity of 0.9 and is maintained at 1000 K, while the interior walls of the furnace are black and are at 800 K. Calculate the net rate at which heat is lost from the rod per unit length.arrow_forwardNUMBER 2 A piece of beef steak 10 cm thick will be frozen in the freezer room -25 ° C. This product has a moisture content of 73%, a density of 970 kg / m³, and a thermal conductivity (frozen) of 1.1 W / (m K). Estimate the freezing time. using the Plank equation. This product has an initial freezing temperature of -1.75 ° C, and the movement of air in the freezing room gives a convective heat transfer coefficient of 15 W / (m² K). t f = hourarrow_forward
- A brass plate has a circular hole whose diameter is slightly smaller than the diameter of an aluminum ball. Ifthe ball and the plate are always kept at the same temperature,(a) should the temperature of the system be increased or decreasedin order for the ball to fit through the hole? (b) Choose the bestexplanation from among the following:I. The aluminum ball changes its diameter more with temperature than the brass plate, and therefore the temperatureshould be decreased.II. Changing the temperature won’t change the fact that the ballis larger than the hole.III. Heating the brass plate makes its hole larger, and that willallow the ball to pass through.arrow_forward(a) The heating power generated by the nuclear heating rod and the inner temperature of the metallic cylinder, T2. (b) The temperature liquid X, which is located between the nuclear heating rod and the meatllic cylinder, Tliquid. (c) The temperature at the surface of the heating rod, T1. (d) If the salt solution temperature is 300 degree Celcius, what is the value of convection heat transfer coefficient, h of the salt solution ? (e) If the heating rod is not covered by metallic cylinder and corresponds to the result of part (c) and (d), how much the rate of energy transfer to the salt solution ? (f) Compare and discuss the difference of heat transfer with and without the matallic cylinder.arrow_forward8. The shown 2-D plate is in contact with a heat source at its upper edge, which supplies heat at a constant flux, qo, per unit length. -- a. Derive a finite-difference relationship to express the steady-state temperature at the shown boundary point TP, in terms of the temperatures at the surrounding points (TE, Tw, Ts) and the other quantities in the problem (e.g., k, qo, etc.). Follow the methodology outlined in the class notes (i.e., use energy balance). Assume Ax = Ay. y 90 b. Modify the relationship for the case when the upper edge is perfectly insulated (without heat addition).arrow_forward
- A square piece is initially at 10 degrees Celsius everywhere. Each side is 0.05 m long. Then the four sides are instantly heated up to 25 degrees Celsius, 50 degrees Celsius, 30 degrees Celsius, and 0 degrees Celsius. a. Find how long it takes for the temperature profile of this square to become steady if the material is copper. What is the final temperature at the center of the square? b. Find how long it takes for the temperature profile of this square to become steady if the material is air. What is the final temperature at the center of the square? c. Which material takes longer to reach steady state and why?arrow_forwardStainless steel (AISI 304) ball bearings, which have been uniformly heated to 850°C, are hardened by quenching them in an oil bath that is maintained at 40°C. The ball diameter is 12.5 mm, and the convection coefficient associated with the oil bath is 1000 W/m².K. (a) If quenching is to occur until the surface temperature of the balls reaches 100°C, how long, in s, must the balls be kept in the oil? What is the center temperature, in °C, at the conclusion of the cooling period? (b) If 10,000 balls are to be quenched per hour, what is the rate at which energy must be removed, in kW, by the oil bath cooling system in order to maintain its temperature at 40°C? Evaluate the properties of the stainless steel (AISI 304) at 500 K. Part A If quenching is to occur until the surface temperature of the balls reaches 100°C, how long, in s, must the balls be kept in the oil? What is the center temperature, in °C, at the conclusion of the cooling period? t = T₁ = i i S °℃arrow_forwardConsider a solid sphere of radius R with a fixed surface temperature, TR. Heat is generated within the solid at a rate per unit volume given by q = ₁ + ₂r; where ₁ and ₂ are constants. (a) Assuming constant thermal conductivity, use the conduction equation to derive an expression for the steady-state temperature profile, T(r), in the sphere. (b) Calculate the temperature at the center of the sphere for the following parameter values: R=3 m 1₁-20 W/m³ TR-20 °C k-0.5 W/(m K) ₂-10 W/m³arrow_forward
- Q1 Passage of an electric current through a long conducting rod of radius r; and thermal conductivity k, results in uniform volumetric heating at a rate of ġ. The conduct- ing rod is wrapped in an electrically nonconducting cladding material of outer radius r, and thermal conduc- tivity k, and convection cooling is provided by an adjoining fluid. Conducting rod, ġ, k, 11 To Čladding, ke For steady-state conditions, write appropriate forms of the heat equations for the rod and cladding. Express ap- propriate boundary conditions for the solution of these equations.arrow_forwardNUMBER 4 A food product wants to be produced in a small round shape (pellet) by freezing it in a water blast freezer freezer. Air freezer operates at -30 ° C. The initial product temperature is 25 ° C. The pellet has a diameter of 0.5 cm, and a density of 980 kg / m³. The initial freezing temperature is -2.5 ° C. The latent heat of freezing of the product is 280 kJ / kg. The thermal conductivity of the frozen product is 1.9 W / (m ° C). The convective heat transfer coefficient is 50 W / (m² K). Calculate the freeze time. t f = hour.arrow_forwardFollowing the Siebel's equation, what is the specific heat of a material with 85% moisture content? If the same material with a mass of 15kg is placed and heated inside a room with temperature of 25 deg Celcius, a resulting 45 deg Celsius change was observed in 15 minutes time. What is the specific heat, the total amount of heat absorbed and the rate of heat absorption of the material considering a linear case scenario? ASAP Answer onlyarrow_forward
 Principles of Heat Transfer (Activate Learning wi...Mechanical EngineeringISBN:9781305387102Author:Kreith, Frank; Manglik, Raj M.Publisher:Cengage Learning
Principles of Heat Transfer (Activate Learning wi...Mechanical EngineeringISBN:9781305387102Author:Kreith, Frank; Manglik, Raj M.Publisher:Cengage Learning
