
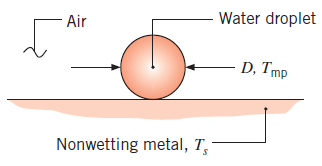
A small water droplet of diameter
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Introduction to Heat Transfer
- At a certain location during the winter, the average air temperature is 10 degrees Celsius and the net radiation is 40 W/m^2 and during the summer the net radiation is 200 W/m^2 and the temperature is 25 degrees Celsius. Compute the evaporation rates using the PRiestley-Taylor Methodarrow_forward800°C and h = 20 W/m².K. Determine A thick oak wall, initially at 25°C, is suddenly exposed to combustion products for which T the time of exposure required, in s, for the surface to reach the ignition temperature of 400°C. Evaluate the properties of the oak (cross grain) at 300 K. t = i S =arrow_forwardfind the driving pressure for a stack that has a height of 18 m and carry a hot gas mixture with an average density of 1.2 k/m3. If you know that the total heat rejection by the stack is 1450 KJ and the c, of the hot gas is 1.8 KJ/Kg.K while the hot gas inlet and outlet temperature are 650 K and 500 K respectively. Assume the hot gas pressure as 2.3 bar while the ambient pressure is 1 bar.arrow_forward
- A wood stove is used to heat a single room. The stove is cylindrical in shape, with a diameter of D = 0.400 m and a length of L = 0.500 m, and operates at a temperature of T, = 200 °C. (a) If the temperature of the room is T, = 20°C, determine the amount of radiant energy delivered to the room by the stove each second if the emissivity of the stove is e = 0.920. (b) By definition, the R-value of a conducting slab is given by Atot(Th – To) Poond R = where Atot is the total surface area, Pcond is the power loss by conduction through the slab, Th and Te are the temperatures on the hotter and cooler sides of the slab. If the room has a square shape with walls of height H = 2.40 m and width W = 7.60 m, determine the R-value of the walls and ceiling required to maintain the room temperature at T = 20°C if the outside temperature is T, = 0°C. Note that we are ignoring any heat conveyed by the stove via convection and any energy lost through the walls and windows via convection or radiation.arrow_forwardCooking an egg involves the denaturation of a protein called albumen. The time required to achieve a particular degree of denaturation is inversely proportional to the rate constant for the process. This reaction has a high activation energy, E, = 418 kJ mol. Calculate how long it would take to cook a traditional three-minute egg on top of mountain peak on a day when the atmospheric pressure there is 355 mmHgarrow_forward2. Calculate the mass of silver initially at 445 °C which would be needed to be dropped into a 435 mL sample of water initially at 23.0 °C in an insulated container to raise the temperature of the water to 33.5 °C. The specific heat capacity of water is 4.184 J K-¹ g¹ and the molar heat capacity of silver is 25.9 J K¹ mol¹¹. 2 Tot 21arrow_forward
- A steel ball with a diameter of 10 mm is taken suddenly from a medium at a temperature of 25ºc and annealed for 20 minutes in the furnace host at a temperature of 800ºc. How many ° C is the temperature of the steel ball when it comes out of the oven? (ρ(steel) = 7854 kg / m3, C(steel) = 434 J / kgK, h(air) = 10 W / m2K and h(oil) =100 W / m2K) a. 705.65 b. 662.90 c. 706.25 d. 750.81 e. 750.20arrow_forwardVery pure liquid water can be subcooled at atmospheric pressure to temperatures well below 0 °C. Assume that 1 kg has been cooled as a liquid to -6 °C. A small ice crystal (of negligible mass) is added to “seed” the subcooled liquid. If the subsequent change occurs adiabatically at atmospheric pressure, what fraction of the system freezes and what is the final temperature? What is ΔStotal for the process, and what is its irreversible feature? The latent heat of fusion of water at 0 °C is 333.4 J/g, and the specific heat of subcooled liquid water is 4.226 J/g °C.arrow_forwardSample Problem • Four metric tons of pork are to be stored at a temperature of -12°C in 8 hours. The product enters the chiller at a temperature of 21°C and the specific heats above and below freezing are 0.86 kCal/kg-°C and 0.51 kcal/kg-°C, respectively, and its latent heat of fusion is 48 kCal/kg. If the freezing temperature of the product is -5°C, determine the product load in kCal/hr. TCLarrow_forward
- Six cylindrical core plugs of 2.54 cm diameter and 3.81 cm length were taken from an Alaskan North Slope reservoir. After cleaning, porosities of all the plugs were measured by a helium porosimeter. Subsequently, all samples were fully saturated with a 0.07 Ω m brine such that Sw = 1. Each sample was placed in a resistivity apparatus, and ΔV values were measured for a current flow of 0.01 A. Determine the formation factor F, for each core plug, and estimate parameters a and m for Archie’s formation factor equation. Core Plug ϕ (Fraction) ΔV (V) 1 0.175 1.72 2 0.19 1.45 3 0.165 1.99 4 0.23 1.1 5 0.16 2.15 6 0.15 2.45arrow_forwardNumber 4 A food product wants to be produced in a small round shape (pellet) by freezing it in a water blast freezer freezer. Air freezer operates at a temperature of -35 ° C. The initial product temperature is 25 ° C. The pellet has a diameter of 1.2 cm, and a density of 980 kg / m³. The initial freezing temperature is -2.5 ° C. The latent heat of freezing of the product is 280 kJ / kg. The thermal conductivity of the frozen product is 1.9 W / (m ° C). The convective heat transfer coefficient is 50 W / (m² K). Calculate the freeze time. identify : a. tf = Answer in hourarrow_forwardQuestion 5: Z=62 a. An iron sphere of mass (Z + 300)g is kept in a container having boiling water (100 °C). If the temperature of the sphere is 25.5°C, how much heat energy is absorbed by the iron sphere? Consider the specific heat of iron as 452J/kg. b. The wall of an industrial furnace is constructed from (Z + 3) cm thick fireclay brick having a thermal conductivity of 1.7 W/mK. Measurements made during steady-state operation reveal temperatures of 530°C and 375°C at the inner and outer surfaces, respectively. Find the rate of heat loss through a wall which is (Z + 5) cm by (Z + 3) m on a side.arrow_forward
 Principles of Heat Transfer (Activate Learning wi...Mechanical EngineeringISBN:9781305387102Author:Kreith, Frank; Manglik, Raj M.Publisher:Cengage Learning
Principles of Heat Transfer (Activate Learning wi...Mechanical EngineeringISBN:9781305387102Author:Kreith, Frank; Manglik, Raj M.Publisher:Cengage Learning
