
(a)
Interpretation:
The IUPAC name of alcohol given in the structure needs to be determined.
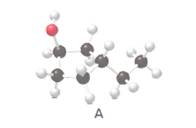
Concept Introduction:
Organic compounds are the compounds which are mainly composed C and H atoms. The branch of chemistry that deals with preparation, reactions, and properties of organic compounds. The molecular formula of organic compound represents the number of bonded atoms with their atomic symbols.
(b)
Interpretation:
The alcohol needs to be classified as 1°, 2°, and 3° alcohol in the given structure.
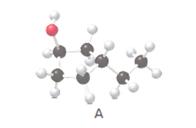
Concept Introduction:
Organic compounds are the compounds which are mainly composed C and H atoms. The branch of chemistry that deals with preparation, reactions, and properties of organic compounds. The molecular formula of organic compound represents the number of bonded atoms with their atomic symbols.
Functional groups are the groups of atoms or atoms which are bonded with parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In organic chemistry, there are different functional groups such as carboxylic acid, alcohol, ester, or amide.
(c)
Interpretation:
The product formed after the dehydration of A with
Concept Introduction:
Organic compounds are the compounds which are mainly composed C and H atoms. The branch of chemistry that deals with preparation, reactions, and properties of organic compounds. The molecular formula of organic compound represents the number of bonded atoms with their atomic symbols.
Functional groups are the groups of atoms or atoms which are bonded with parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In organic chemistry, there are different functional groups such as carboxylic acid, alcohol, ester, or amide.
(d)
Interpretation:
The product that is formed when 3-ethylcyclopentanol (A) is oxidized with K2Cr2O7 needs to be determined.
Concept Introduction:
Organic compounds are the compounds which are mainly composed C and H atoms. The branch of chemistry that deals with preparation, reactions, and properties of organic compounds. The molecular formula of organic compound represents the number of bonded atoms with their atomic symbols.
Functional groups are the groups of atoms or atoms which are bonded with parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In organic chemistry, there are different functional groups such as carboxylic acid, alcohol, ester, or amide.
(e)
Interpretation:
The constitutional isomer of 3-ethylcyclopentanol that contains one −OH group needs to be determined.
Concept Introduction:
Organic compounds are the compounds which are mainly composed C and H atoms. The branch of chemistry that deals with preparation, reactions, and properties of organic compounds. The molecular formula of organic compound represents the number of bonded atoms with their atomic symbols.
Functional groups are the groups of atoms or atoms which are bonded with parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In organic chemistry, there are different functional groups such as carboxylic acid, alcohol, ester, or amide.
(f)
Interpretation:
The constitutional isomer of 3-ethylcyclopentanol that contains an ether group needs to be determined.
Concept Introduction:
Organic compounds are the compounds which are mainly composed C and H atoms. The branch of chemistry that deals with preparation, reactions, and properties of organic compounds. The molecular formula of organic compound represents the number of bonded atoms with their atomic symbols.
Functional groups are the groups of atoms or atoms which are bonded with parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In organic chemistry, there are different functional groups such as carboxylic acid, alcohol, ester, or amide.
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Which alkyl halide has the highest boiling point? A. CH3BrB. CH3FC. CH3ClD. CH3larrow_forwardGlycerol contains: a. oxygens which are each bonded to two alkyl groups b. oxygens single-bonded to primary and secondary carbons c. Oxygens double-bonded to carbon, with alkyls on both sides d. Oxygens double-bonded to carbon, with alkyls on one side only e. Oxygens double-bonded to carbon, with an alkyl on one side and an --OH on the other sidearrow_forwardALCOHOLS 1. WHY IS ETHANOL MORE SOLUBLE IN WATER THAN 1-HEXANOL? 2. WHAT IS DENATURED ALCOHOL? AND WHY IS ALCOHOL DENATURED? ETHER 1. WHY DOES DIETHYL ETHER HAVE MUCH LOWER BOILING POINT THAN 1-BUTANOL?arrow_forward
- The correct IUPAC name for this compound is: O a. 4-oxa-1-cyclohexanol O b. 4-hydroxyoxacyclohexene O c. 4-oxa-1-cyclohexenol O d. 4-oxacyclohexane-1-ol OHarrow_forward1. When an aldehyde is reduced with hydrogen (H2) gas in the presence of a transition metal catalyst, what type of product is formed? A. primary alcohol B. secondary alcohol C. tertiary alcohol D. carboxylic acidarrow_forwardWhat is produced when methanal undergoes Grignard reaction? O A. primary alcohol B. secondary alcohol O C. tertiary alcohol O D. no reactionarrow_forward
- 3. Complete the following intramolecular dehydration reactions for alcohols. Draw the structure of the product. Name the reactant and the product. a) Cy-C-CH-oH 18UC b) CHy CH-Cy-CHy 180C OH c) 180c 4. Complete the following intermolecular dehydration reactions for alcohols. Draw the structure of the product. Name the reactant and the product. a) CHy-CH-OH b) Ho SわふわSわ 5. Complete the following oxidation reactions for alcohols. Draw the structure of the product. Name the reactant and identify the type of compound formed in the product. a) hparrow_forwardDraw the structure of an alkane that: a. Contains only 1° and 4° carbons. c. Contains only 1° and 2° hydrogens. b. Contains only 2° carbons. d. Contains only 1° and 3° hydrogens.arrow_forward7 Give the IUPAC name for the following compound. OH A. 3-methyl-1-pentenyl alcohol B. 3-vinyl-1-butanol C. 3-methyl-4-penten-1-olarrow_forward
- A Primary alcohol will get converted into what functional group in the following reaction? 1. DMSO, (COCI)2 Primary Alcohol ???? 2. EtgN carboxylic acid ketone aldehyde ester D. A Darrow_forward2. Complete the following reactions for the preparation of alcohols. Draw the structure of the product. Name the reactant and the product. a) CHy CH=CH-CHy + HyO Hydration of alkenes b) Cat. CHy-CHy + H2 drogenation carbonyl pups d)arrow_forwardIdentify the IUPAC name of the given structure. A. 2 - methylhexan-5-one B. 5 - methylhexan-2-one C. 2 - heptanone D. 5 - heptanone Identify the IUPAC name of the given structure. A. 4 - bromopentan-3-one B. 1 - bromobutan-2-one C. 2 - bromobutan-one D. None of the abovearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





