
General, Organic, and Biological Chemistry - 4th edition
4th Edition
ISBN: 9781259883989
Author: by Janice Smith
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Question
Chapter 14, Problem 33P
Interpretation Introduction
(a)
Interpretation:
The name of the following molecule which is depicted in the ball-and-stick model should be determined.
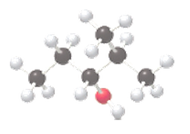
Concept Introduction:
Name of the compound can be assigned according to below rule:
First, identify the number of carbon atoms present in the longest continuous chain, and then identify the functional group if present in the given compound.
Interpretation Introduction
(b)
Interpretation:
The name of the following molecule which is depicted in the ball-and-stick model should be determined.
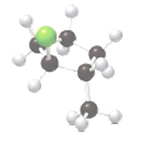
Concept Introduction:
Name of the compound can be assigned according to below rule:
First, identify the number of carbon atoms present in chain, and then identify the
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Explain why these compounds (1-propanol, ethylene glycol, and ethanethiol) experience different intermolecular forces based on their molecular structure.
Name each compound depicted in the ball-and-stick models.
Cyclopentane has a higher boiling point than pentane even though both compounds have the same number of carbons (49 °C vs. 36 °C). Can you suggest a reason for this phenomenon?
Chapter 14 Solutions
General, Organic, and Biological Chemistry - 4th edition
Ch. 14.1 - Prob. 14.1PCh. 14.2 - Prob. 14.1PPCh. 14.2 - Classify each hydroxyl group in sorbitol as 1°,...Ch. 14.2 - Prob. 14.3PCh. 14.2 - Prob. 14.4PCh. 14.3 - Prob. 14.2PPCh. 14.3 - Give the structure corresponding to each name a....Ch. 14.5 - Draw the products formed when each alcohol is...Ch. 14.5 - Prob. 14.6PCh. 14.5 - Prob. 14.7P
Ch. 14.5 - Prob. 14.4PPCh. 14.6 - Prob. 14.8PCh. 14.7 - Prob. 14.9PCh. 14.7 - Prob. 14.5PPCh. 14.7 - Prob. 14.10PCh. 14.7 - Name each ether. CH3OCH2CH2CH2CH3Ch. 14.7 - Prob. 14.11PCh. 14.8 - (a) Translate the hall and stick model of...Ch. 14.8 - Prob. 14.13PCh. 14.9 - Prob. 14.14PCh. 14.9 - Prob. 14.15PCh. 14.9 - Prob. 14.16PCh. 14.9 - Prob. 14.17PCh. 14.9 - Prob. 14.18PCh. 14.9 - Prob. 14.19PCh. 14.9 - Prob. 14.20PCh. 14.10 - Give the IUPAC name for each thiol.Ch. 14.10 - Prob. 14.21PCh. 14.10 - Prob. 14.22PCh. 14 - Classify each alcohol as 1°, 2°, or 3o a....Ch. 14 - Prob. 24PCh. 14 - Prob. 25PCh. 14 - Classify each halide hi A as 1°, 2°, or 3°. A is a...Ch. 14 - Prob. 27PCh. 14 - Draw the structure of a molecule that fits each...Ch. 14 - Draw the structure of the six constitutional...Ch. 14 - Draw the structure of the four constitutional...Ch. 14 - Draw the structure of l-propanethiol, a compound...Ch. 14 - Prob. 32PCh. 14 - Prob. 33PCh. 14 - Prob. 34PCh. 14 - Prob. 35PCh. 14 - Answer each question about alcohol B. Draw a...Ch. 14 - Prob. 37PCh. 14 - Give the IUAPC name for each alcohol.Ch. 14 - Prob. 39PCh. 14 - Prob. 40PCh. 14 - Prob. 41PCh. 14 - Prob. 42PCh. 14 - Draw the structures and give the IUPAC names for...Ch. 14 - Prob. 44PCh. 14 - Prob. 45PCh. 14 - Prob. 46PCh. 14 - Give the structure corresponding to each IUPAC...Ch. 14 - Give the structure corresponding to each IUPAC...Ch. 14 - Which compound in each pair has the higher boiling...Ch. 14 - Rank the compounds in order of increasing melting...Ch. 14 - Rank the following compounds in order of...Ch. 14 - Rank the following compounds in order of...Ch. 14 - Prob. 53PCh. 14 - Prob. 54PCh. 14 - Prob. 55PCh. 14 - Prob. 56PCh. 14 - Prob. 57PCh. 14 - Prob. 58PCh. 14 - Prob. 59PCh. 14 - Prob. 60PCh. 14 - Prob. 61PCh. 14 - Prob. 62PCh. 14 - Prob. 63PCh. 14 - Prob. 64PCh. 14 - Prob. 65PCh. 14 - Prob. 66PCh. 14 - Prob. 67PCh. 14 - Prob. 68PCh. 14 - Prob. 69PCh. 14 - Prob. 70PCh. 14 - Prob. 71PCh. 14 - Prob. 72PCh. 14 - Prob. 73PCh. 14 - Prob. 74PCh. 14 - Prob. 75PCh. 14 - Prob. 76PCh. 14 - Prob. 77PCh. 14 - Prob. 78PCh. 14 - Prob. 79PCh. 14 - Prob. 80PCh. 14 - With reference to the halogenated organic...Ch. 14 - Prob. 82PCh. 14 - Prob. 83PCh. 14 - Prob. 84PCh. 14 - Write out the chemical reaction that occurs when a...Ch. 14 - Prob. 86PCh. 14 - Prob. 87PCh. 14 - Lactic acid [CH3CH(OH)CO2H] gives sour milk its...Ch. 14 - Prob. 89PCh. 14 - Prob. 90PCh. 14 - Prob. 91PCh. 14 - Prob. 92PCh. 14 - Prob. 93PCh. 14 - Answer the following questions about alcohol B....Ch. 14 - Prob. 95CPCh. 14 - Dehydration of alcohol C forms two products of...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Give the common name for NN OH T CH3-CH₂-C-N-CH₂-CH3 Spell out the common name of the compound. Give the IUPAC name for 0 CH₂ || CH₂-C-N-CH₂-CH₂-CH₂, Spell out the IUPAC name of the compound. diethylamine Draw all missing reactants and/or products in the appropriate boxes by placing atoms on the canvas and connecting them with bonds. Add charges where needed. HE CONT HOarrow_forwardEncircle the functional groups, label each functional group with a letter, and identify the type or class of compounds represented by each functional group. 1. LSD, a hallucinogenic drug widely believed to be the inspiration behind the Beatles hit “Lucy in the Sky with Diamonds” 2. Methyljasmonate, a compound that belongs to a group called the pheromones. The male oriental fruit moth, Grapholitha molesta Busk., responds when a female moth emits this compound. 3.arrow_forwardClassify each molecule as an aldehyde, ketone, or neither. AND Classify each molecule as an ester, ether, or neither.arrow_forward
- Explain the solubility of alcohols in water and in n-hexane.arrow_forwardWhat is the IUPAC name for the compound shown? Step 1: Identify the number of carbons in the longest chain Step 2: Identify the base name of the molecule. Step 3: Number the longest chain. Step 4: Identify substituents. Step 5: Order the substituents. Step 6: Add the substituent locants or numbering. Step 7: Put it all together and give the IUPAC name. Step 7: Put it all together and give the IUPAC name. You have determined the name of the longest carbon chain and the locations and name of the substituents. Complete the name of the molecule by adding in the hyphens and commas. 3ethyl223trimethyl5propyldecane 3-ethyl,2,2,3trimethyl-5propyldecane IUPAC name: Incorrectarrow_forwardWhat is the IUPAC name for the compound shown? Step 1: Identify the number of carbons in the longest chain Step 2: Identify the base name of the molecule. Step 3: Number the longest chain. Step 4: Identify substituents. Step 5: Order the substituents. Step 6: Add the substituent locants or numbering. Step 7: Put it all together and give the IUPAC name. Step 7: Put it all together and give the IUPAC name. You have determined the name of the longest carbon chain and the locations and name of the substituents. Complete the name of the molecule by adding in the hyphens and commas. 3ethyl223trimethyl5propyldecane JUPAC name:arrow_forward
- Name the following alcohols utilizing the IUPAC nomenclature rules a. CH3 CH3-CH,-C–CH2–CH,–CH3 ОН b. CH; —CH, —СН— СН, — СН; CH —ОН с. CH3 CH3- -ОНarrow_forwardGive the IUPAC name for the chemical in the attached image. [click on attachment to view image if you don't see it] CH3CH₂CH₂CH₂CHCH₂CH₂CH₂CH3 CH3CH₂CH₂ O A. 2,2,5,6-tetraethylhexane B. 2,2,5-triethyloctane O C. 3,6-diethyl-3-methylnonane CH₂CH3 D. 3-ethyl-3-methyl-6-propyldecane CH3arrow_forwardDraw the structures of two specific ethers that have the formula C4H10Oarrow_forward
- What is the IUPAC name for the compound shown? Step 1: Identify the number of carbons in the longest chain Step 2: Identify the base name of the molecule. Step 3: Number the longest chain. Step 4: Identify substituents. Step 5: Order the substituents. Step 6: Add the substituent locants or numbering. Step 7: Put it all together and give the IUPAC name. Step 3: Number the longest chain of carbon atoms so that the substituents have the lowest possible numbers. Carbon atoms that are not in the longest chain should not be numbered. Answer Bank 10 5 3 8 2.arrow_forwardWhat is the IUPAC name for the compound shown? Step 1: Identify the number of carbons in the longest chain Step 2: Identify the base name of the molecule. Step 3: Number the longest chain. Step 4: Identify substituents. Step 5: Order the substituents. Step 6: Add the substituent locants or numbering. Step 7: Put it all together and give the IUPAC name. Step 5: Order the substituents. Since there are multiple substituents present, we must next consider the order the substituents in the name. The substituent order should be methyl, ethyl, followed by propyl ethyl, propyl, followed by trimethyl trimethyl, ethyl followed by propyl O ethyl, trimethyl, followed by propylarrow_forwardWhat is the IUPAC name for the compound shown? Step 1: Identify the number of carbons in the longest chain Step 2: Identify the base name of the molecule. Step 3: Number the longest chain. Step 4: Identify substituents. Step 5: Order the substituents. Step 6: Add the substituent locants or numbering. Step 7: Put it all together and give the IUPAC name. Step 6: Add the substituent locants or numbering. Now that you have identified the order of the substituents, you need to provide the locant, which indicates the number carbon on the main chain where each substituent is placed. Carbon 5 has only one substituent, so the name is represented by 5-propyl. Both carbons 2 and 3 have two substituents. What would be the correct naming for those carbons? The substituents should be named: 2,2-dimethyl and 3-methyl-3-ethyl 2,3-trimethyl and 3-ethyl 2,2,3-trimethyl and 3-ethyl 2-dimethyl and 3-methylethylarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

07 Physical Properties of Organic Compounds; Author: Mindset;https://www.youtube.com/watch?v=UjlSgwq4w6U;License: Standard YouTube License, CC-BY