
(a)
Interpretation:
An acceptable name for the following ether should be determined:
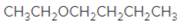
Concept Introduction:
Ethers are molecules which have two alkyl groups bonded to the oxygen atom.
Simple ethers named based on their structure.
When ether nomenclature, both alkyl groups are named and arranged them according to the alphabetical order and at the end of the name word 'ether' should be added. Ethers which have identical alkyl groups, prefix di- should be added to the alkyl group.
When naming more complex ethers according to the
(b)
Interpretation:
An acceptable name for the following ether should be determined:
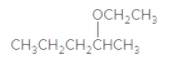
Concept Introduction:
Ethers are molecules which have two alkyl groups bonded to the oxygen atom.
Simple ethers named based on their structure.
When ether nomenclature, both alkyl groups are named and arranged them according to the alphabetical order and at the end of the name word 'ether' should be added. Ethers which have identical alkyl groups, prefix di- should be added to the alkyl group.
When naming more complex ethers according to the IUPAC nomenclature; one alkyl group is named as a hydrocarbon chain and the other part is named as a part of substituent bonded to that chain. The simpler alkyl group with the oxygen atom should name as an alkoxy substituent and the -yl suffix of the alkyl replaced with- oxy suffix. Remaining longest alkyl chain is named as an alkane by considering the alkoxy group as a substituent bonded to the main carbon chain.
(c)
Interpretation:
An acceptable name for the following ether should be determined:
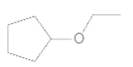
Concept Introduction:
Ethers are molecules which have two alkyl groups bonded to the oxygen atom.
Simple ethers named based on their structure.
When ether nomenclature, both alkyl groups are named and arranged them according to the alphabetical order and at the end of the name word 'ether' should be added. Ethers which have identical alkyl groups, prefix di- should be added to the alkyl group.
When naming more complex ethers according to the IUPAC nomenclature; one alkyl group is named as a hydrocarbon chain and the other part is named as a part of substituent bonded to that chain. The simpler alkyl group with the oxygen atom should name as an alkoxy substituent and the -yl suffix of the alkyl replaced with- oxy suffix. Remaining longest alkyl chain is named as an alkane by considering the alkoxy group as a substituent bonded to the main carbon chain.
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
General, Organic, and Biological Chemistry - 4th edition
- 1. Name the following ether. 2. Name the following thiol. C C-C-C-C-0-C-C -9-9-⁹ C-C-C-C-C-S-H C-C-C-C 3. Will the ether H bond with water? Will the thiol H bond with water?arrow_forwardGive the IUPAC name for each compound. Part 1 of 3 X S 1arrow_forwardName the following alcohol. O2-Bromo-4-ethylcyclopentanol 4-Ethyl-2-bromocyclopentanol O1-Ethyl-3-bromo-4-cyclopentanol O1-Bromo-4-ethyl-2-cyclopentanolarrow_forward
- Give both the IUPAC name and the common name of the ether shown. Part 1 of 2 IUPAC name: Part 2 of 2 Common name: X X Ś 5arrow_forwardPredict which member of each group is most soluble in water, and explain the reasons for your predictions.chlorocyclohexane, cyclohexanol, or cyclohexane-1,2-diolarrow_forwardHCI CH3OH CH₁₂arrow_forward
- Give the IUPAC name for each structure. Part 1 of 3 CH,(CH,),CO,CH, 0 X $arrow_forwardGive the IUPAC name for each compound. Part 1 of 3 CH₂CH, H,C-C-CH_CH, CH, 1 H Part 2 of 3 X CH, H H,CCH,CH,-с-с-сH CH CH,CH, I H CH₂CH₂CH, Part 3 of 3 X H CH₂CH₂CH, H,CCH, -e-c-CH,CH, H,CCH, Йarrow_forwardexplain why methanethiol, CH3SH, has a lower boiling point (6°C) than methanol, CH3OH (65°C), even though methanethiol has a higher molecular weightarrow_forward
- Show how each alcohol or diol can be prepared from an alkene. (a) 2-Pentanol (b) 1-Pentanol (c) 2-Methyl-2-pentanol (d) 2-Methyl-2-butanol (e) 3-Pentanol (f) 3-Ethyl-3-pentanol (g) 1,2-Hexanediolarrow_forwardDraw the eight constitutional isomers with molecular formula C5H120 that contain an OH group. Give the IUPAC name for each compound. Classify each alcohol as 1°, 2°, or 3°.arrow_forwardCH3 OH HBr ? ether OH PBr3 etherarrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

