
Concept explainers
(a)
Interpretation:
The product formed from the condensation reaction of propanoic acid and 2-methylpropanol has to be drawn.
Concept Introduction:
Ester:
The structure of an ester is,
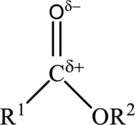
The carbonyl group of the ester is polar and could participate in dipole-dipole attractions. Hydrocarbon chains flank the carbonyl group
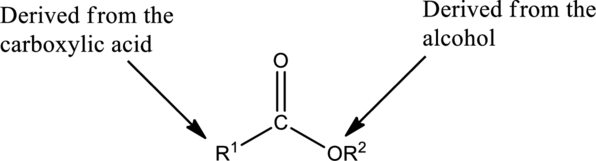
Preparation of Esters:
Esters are prepared from the reaction of
The general preparation of esters is shown below,
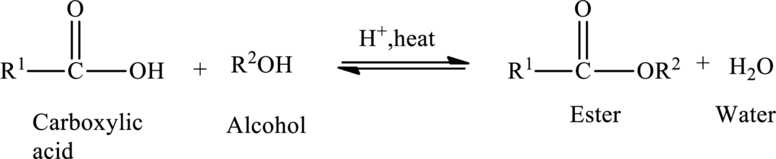
(b)
Interpretation:
The product formed from the condensation reaction of ethanoic acid and cyclohexanol has to be drawn.
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
The product formed from the condensation reaction of butanoic acid and dimethyl
Concept Introduction:
Amides can be prepared in a reaction between a carboxylic acid and an amine. The acid derivative can be either an acid chloride or an acid anhydride.
The general reaction is,
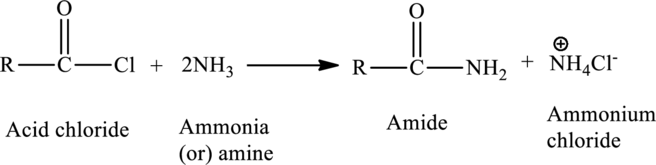
(d)
Interpretation:
The product formed from the condensation reaction of 2-methylpentanoic acid and ethylamine has to be drawn.
Concept Introduction:
Refer to part (d).
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
Chemical Principles: The Quest for Insight
- Write the equations for the preparation of alcohols by reacting a) propene with sulfuric acid and water b) chlorocyclohexane and sodium hydroxide Write the equations for the reaction of phenoxybenzene with hot concentrated HCIarrow_forwardDraw the organic product formed when the followingcompounds undergo a substitution reaction: (a) acetic acid and1-hexanol; (b) propanoic acid and dimethylamine; (c) ethanoicacid and diethylamine.arrow_forwardDraw the structures of the following compounds:(a) Ethanoic acid(b) Bromopentane(c) Butanonearrow_forward
- The reactant: benzophenone & 2-propanol the product: benzopinacol & acetonearrow_forwardThe foul odor of rancid butter is caused by butyric acid, CH3CH2CH2CO2H.(a) Draw the Lewis structure and determine the oxidation number and hybridization for each carbon atom in the molecule.(b) The esters formed from butyric acid are pleasant-smelling compounds found in fruits and used in perfumes. Draw the Lewis structure for the ester formed from the reaction of butyric acid with 2-propanol.arrow_forwardGive the structural formulae and name the functional groups of the following compounds. (a) 3-chlorobut-1-ene Name the functional group: (b) butanedioic acid Name the functional group: (c) propanamide Name the functional group: (d) 3-methylbutanal Name the functional group:arrow_forward
- Draw the product resulting from mild oxidation of (a) 2-butanol; (b) 2-methylpropanal; (c) cyclopentanol.arrow_forward5. Give the structural formulae and name the functional groups of the following compounds. (a) 3-chlorobut-1-ene (b) butanedioic acid Name the functional group: (c) propanamide Name the functional group: (d) 3-methylbutanal Name the functional group: Name the functional group:arrow_forwardSynthesize each compound from benzonitrile (C6H5CN) as the only organic starting material; that is, every carbon in the product must originate in benzonitrile.arrow_forward
- Which of the isomeric C4H₁0O alcohols can be prepared by hydrogenation of aldehydes? Which can be prepared by hydrogenation of ketones? Which cannot be prepared by hydrogenation of a carbonyl compound?arrow_forwardDraw the structure and name the product formed if the following alcohols are oxidized. Assume an excess of the oxidizing agent is used. If the alcohol is not expected to react with a chemical oxidizing agent, write NR (no reaction).(a) CH3CH2CH2CH2OH(b) 2-butanol(c) 2-methyl-2-propanol(d) 2-methyl-1-propanolarrow_forwardDiazomethane, CH2N2, is used in the organic chemistry laboratory despite its danger because it produces very high yields and is selective for reaction with carboxylic acids. Write the products of the following reactions.arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
