
(a)
Interpretation:
The systematic name of the compound
Concept Introduction:
Rules for naming compound:
- 1) The carbon atoms in the longest chain have to be counted.
- 2) The substituents and multiple bonds have to be identified and counted and the suffix “-ene” is added.
- 3) The backbone carbon atoms have to be numbered by assigning the lowest number from the starting end that contains the double bond.
Rules for naming compound:
- 1) The carbon atoms in the longest chain have to be counted.
- 2) The substituents and multiple bonds have to be identified and counted and the suffix “-yne” is added.
- 3) The backbone carbon atoms have to be numbered by assigning the lowest number from the starting end that contains the double bond.
Geometrical isomers: In geometrical isomers, atoms have different arrangements on either side of a double bond above or below the ring of a cycloalkane or cycloalkane. If the atoms are present on the same side of the double bond, then it is cis-isomer and if they are present on the opposite side of the double bond, then it is trans-isomer.
(a)
Answer to Problem 11.8E
The systematic name of the compound
Cis-
Explanation of Solution
The given compound is,
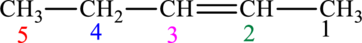
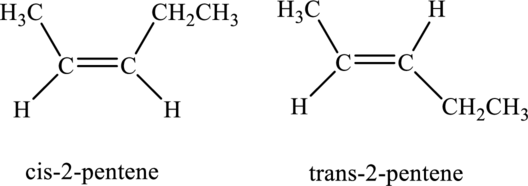
The compound is identified as alkene. The parent chain of the compound is pentane and a double bond is seen at the carbon second position. Hence, the systematic name of the compound is
Cis-
(b)
Interpretation:
The systematic name of the compound
Concept Introduction:
Refer to part (a).
(b)
Answer to Problem 11.8E
The systematic name of the compound
The geometrical isomers are not possible in
Explanation of Solution
The given compound is,
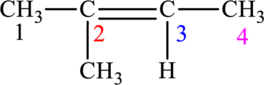
The compound is identified as alkene. The parent chain of the compound is butane. One methyl group is present in the carbon second position and a double bond is seen at the carbon second position. Hence, the systematic name of the compound is
No geometrical isomers are possible in
(c)
Interpretation:
The systematic name of the compound
Concept Introduction:
Refer to part (a).
(c)
Answer to Problem 11.8E
The systematic name of the compound
No geometrical isomers are possible in
Explanation of Solution
The given compound is,
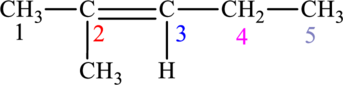
The compound is identified as alkene. The parent chain of the compound is pentane. One methyl group is present in the carbon second position and a double bond is seen at the carbon second position. Hence, the systematic name of the compound is
No geometrical isomers are possible in
(d)
Interpretation:
The systematic name of the compound
Concept Introduction:
Refer to part (a).
(d)
Answer to Problem 11.8E
The systematic name of the compound
No geometrical isomers are possible in
Explanation of Solution
The given compound is,
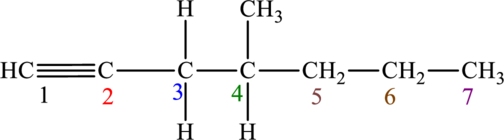
The compound is identified as alkyne. The parent chain of the compound is heptane. One triple bond is present in carbon first position and one methyl substituent is present in carbon fourth position. Hence, the systematic name of the compound is
Geometrical isomers are not possible because triple bond has only one substituent each.
(e)
Interpretation:
The systematic name of the compound
Concept Introduction:
Refer to part (a).
(e)
Answer to Problem 11.8E
The systematic name of the compound
No geometrical isomers are possible in
Explanation of Solution
The given compound is,
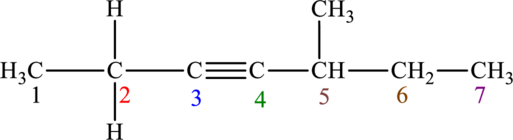
The compound is identified as alkyne. The parent chain of the compound is heptane. One triple bond is present in carbon third position and one methyl substituent is present in carbon fifth position. Hence, the systematic name of the compound is
Geometrical isomers are not possible because triple bond has only one substituent each.
Want to see more full solutions like this?
Chapter 11 Solutions
Chemical Principles: The Quest for Insight
- Draw the structures of the following compounds. (a) 1-isobutyl-4-isopropylcyclohexane; (b) tert-butylcyclopentane;(c) 3,3-diisopropyloctanearrow_forwardDraw structures for the following haloalkanes. (a) 1,2,3-tribromohexane; (b) 2,2,3,3,4-pentachlorohexane;(c) 1,2-dichloro-4-nitrohexane; (d) 1,2-dichloro-3-methoxycyclopentanearrow_forwardWrite the bond line formula of the following compounds: (a) 4-methyl-2-hexene, two geometrical (stereoisomers) isomers (b) 3-fluoro-2-methylheptanol (3-fluoro-2-methylheptan-1-ol) (c) 4-methyl-hex-1-yn-3-olarrow_forward
- Write structural formulas for compounds that meet the following descriptions:(a) An alkene, C6H12, that cannot have cis–trans isomersand whose longest chain is 5 carbons long(b) An alkene with a chemical formula of C10H12 that hascis–trans isomers and contains a benzene ring.arrow_forward5. Starting with propyne and any other reactants, show by equations the synthesis of: (a) 2-bromo-2-chloropropane(b) 1,2-dibromo-2-chloropropane (c) 1,1,2,2-tetrachloropropane 3. The following names are incorrect: State why each name is wrong and give the correct name (a) 3-methyl-3-butene(b) cis-3-pentene(c) cis-2-methyl-2-pentenearrow_forwardIllustrate the chemical structural formula for 3-methyl-3-ethylpentane. (b) Identify its chemical family as an isomer. (c) Provide the balanced chemical reaction equation for the combustion of one mole of this fuel with an equivalence ratio of ϕ=0.735arrow_forward
- Draw the structure(s) of all of the branched alkene isomers, C6H12, that contain 2 methyl branches.arrow_forward1. The structure of compound A is shown below. OH NH2 (a) Redraw the above structure in the form of expanded and condensed structures. (b) Determine the number of primary, secondary, tertiary and quaternary carbon atom that can be found in the compound. (c) Redraw, circle and name the functional groups present in the compound. (d) State the possible type of stereoisomerism of the compound and draw the appropriate structures to describe the isomerism.arrow_forwardProstaglandin F2α, a hormone that causes uterine contraction during childbirth, has the following structure. Are the two hydroxyl groups (-OH) on the cyclopentane ring cis or trans to each other? What about the two carbon chains attached to the ring?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





