
Concept explainers
(a)
Interpretation:
The structural formula of the product of the reaction of glycerol with stearic acid has to be drawn.
Concept Introduction:
Preparation of Esters:
Esters are prepared from the reaction of
The general preparation of esters is shown below,
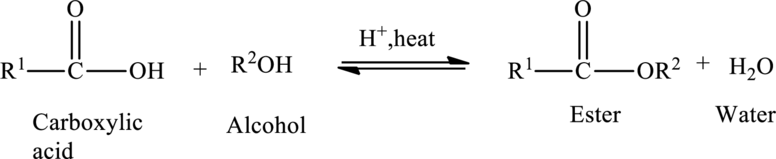
Structural formula: Structural formulas identify the location of
(b)
Interpretation:
The structural formula of the product of the oxidation reaction of 4-hydroxybenzyl alcohol has to be drawn.
Concept Introduction:
The oxidation of a primary alcohol produces an aldehyde.
The generalized equation is written as,
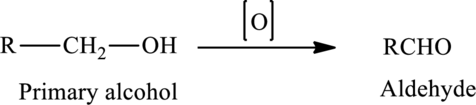
The oxidation of a secondary alcohol produces a ketone.
The generalized equation is written as,
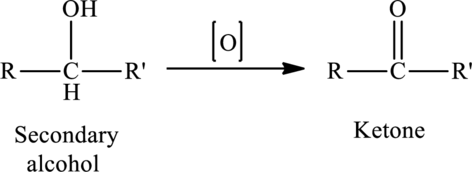
Structural formula: Structural formulas identify the location of chemical bonds between the atoms of a molecule. A structural formula consists of symbols for the atoms connected by short lines that represent chemical bonds-one, two or three standing lines for single, double or triple bonds respectively.
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
Chemical Principles: The Quest for Insight
- Draw the organic product formed when the following compounds undergo a substitution reaction:(a) acetic and 1-hexanol; (b) propanoic acid and dimethylamine; (c) ethanoic acid and dimethylamine.arrow_forwardDraw the organic product formed when the following compounds undergo a substitution reaction: (a) acetic acid and 1-hexanol; (b) propanoic acid and dimethyl-amine; (c) ethanoic acid and diethylamine.arrow_forwardDraw the structure and name the product formed if the following alcohols are oxidized. Assume an excess of the oxidizing agent is used. If the alcohol is not expected to react with a chemical oxidizing agent, write NR (no reaction).(a) CH3CH2CH2CH2OH(b) 2-butanol(c) 2-methyl-2-propanol(d) 2-methyl-1-propanolarrow_forward
- (b) Differentiate between bioethanol and biodiesel.arrow_forwardButanone is a four carbon compound with the functional group:(a) carboxylic acid(b) aldehyde(c) ketone(d) alcoholarrow_forward(i) Draw the structural formula of compounds L, M, N and P (ii) Name the type of chemical reaction for the formation of compound N.arrow_forward
- 5. Give the structural formulae and name the functional groups of the following compounds. (a) 3-chlorobut-1-ene (b) butanedioic acid Name the functional group: (c) propanamide Name the functional group: (d) 3-methylbutanal Name the functional group: Name the functional group:arrow_forwardDraw a structural formula of the product of oxidation of natural rubber by ozone followed by a workup in the presence of (CH3)2S. Name each functional group present in this product.arrow_forwardWhat is the structure of the alcohol produced when 3-methyl-1-pentene undergoes (a) acid catalyzed hydration (b) oxymercuration/demercuration (c) hydroboration/oxidationarrow_forward
- Which of the following describes a chemical property of methanol? (A) It is a colorless liquid; (B) It is a flammable liquid; (C) It is a volatile liquid; (D) It has a low boiling point;arrow_forwardWrite two complete, balanced equations for each of the following reactions, one using condensed formulas and one using Lewis structures:(a) ethanol reacts with propionic acid(b) benzoic acid, C6H5CO2H, is added to a solution of sodium hydroxidearrow_forwardWrite a balanced chemical equation using condensedstructural formulas for (a) the formation of butyl propionatefrom the appropriate acid and alcohol, (b) the saponification(base hydrolysis) of methyl benzoate.arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

