
Concept explainers
a.
Interpretation:
The given pair of compounds as constitutional isomers or identical molecules or not isomers has to be labeled.
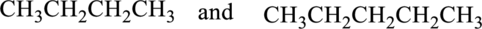
Concept Introduction:
Isomers:
The organic compound contains same molecular formula and different arrangement of atoms is termed as isomers. The isomers have different physical and chemical properties.
Constitutional Isomer:
Constitutional isomer has same molecular formula but they have different connectivity to each other. It is also known as structural isomers.
Identical Molecules:
The compound should have the same number of atoms and has to be arranged in same spatial arrangement is known as identical molecules.
b.
Interpretation
The given compounds as constitutional isomers or identical molecules or not isomers have to be labeled.
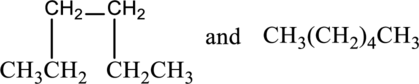
Concept Introduction:
Refer part a.
c.
Interpretation:
The given compounds as constitutional isomers or identical molecules have to be labeled.
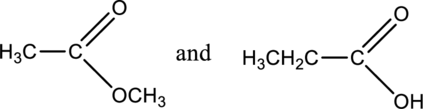
Concept Introduction:
Refer part a.
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
Principles of General, Organic, Biological Chemistry
- Functionalized Hydrocarbons Identify each compound according to its functional group (e.g.,amine,ester,etc.):arrow_forwardCompare the two structural formulas in each set. Do they represent the same compound, represent constitutional isomers or represent two compounds which are neither identical nor constitutional isomers?arrow_forwardidentify each functional groupsarrow_forward
- Beeswax contain approximately 10% hentriacontane, a normal alkane with 31 carbon atoms. What is the molecular formula of hentricontane? Write a completely condensed formula of hentriacontane.arrow_forwardRegarding the three compounds in the previous question which of the following statements is true? Select one: These three compounds are isomers because they have the same molecular formulae. These three compounds are isomers because they have different boiling points. These three compounds are isomers because they have different structures. These three compounds are isomers because they have the same IUPAC name.arrow_forwardThese two compounds represent the same organic compound. CH3 CH3-CH-CH-CH2-CH2-CH3 CH2 CH3arrow_forward
- 4. Draw 10 constitutional isomers for the following molecule. C6H12 alkenearrow_forwardIndicate for the following compound: (1) the number of carbons in the longest carbon chain, (2) the positions of the double bonds [separate # with comma], (3) the IUPAC name of the compound. Answer for blank # 1: 8 Answer for blank # 2: 2,5 Answer for blank # 3: 2,5-dimethylarrow_forward7. Functional isomers have unique functional groups but identical reactivity. true or false?arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning  Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning




