
a.
Interpretation:
The compounds butane or isobutane has to be represented.
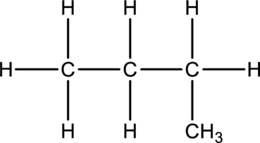
Concept Introduction:
Butane and Isobutane:
In butane structure there will be four carbon atoms present in the straight chain. The carbon present in one continuous chain.
In isobutane there will be three carbon atoms present in a row and one carbon bond is bonded to middle carbon. This
b.
Interpretation
The compounds whether butane or isobutane has to be identified.
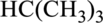
Concept Introduction:
Refer part: a.
c.
Interpretation:
The compounds either butane or isobutane has to be represented.
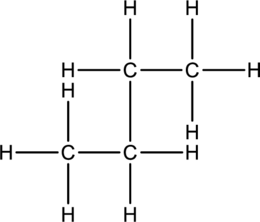
Concept Introduction:
Refer part: a.
d.
Interpretation:
The compounds butane or isobutane has to be represented.
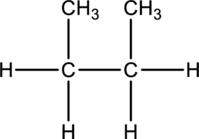
Concept-Introduction:
Refer part: a.
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
Principles of General, Organic, Biological Chemistry
- The octane rating for gasoline is a measurement of how readily a fuelcombusts compared to 2,2,4-trimethylpentane, an isomer of octane.a. Draw 2,2,4-trimethylpentane and verify that it is an isomer ofoctane.b. Draw four other isomers of octane.c. Select one of the isomers and draw it such that it looksdifferent on the page but is still the exact same compound.d. Name this isomer.e. Define isomer using a complete sentence.arrow_forwardName each alkane. b. CH3-CH2-CH-CH, CH, CH3 CH3 CH-CH, c. CH;-CH-CH2-CH-CH;-CH,-CH,arrow_forward3. Name the following alkenes and alkynes. HC-CH-C=C-CH-CH--CH3 CH-CH H H H H. H C= C C C-H H. H.arrow_forward
- What is the preferred IUPAC name of the alkene shown in Figure below? CH3 CH3 CH3 `CH-CH3 A. 1-ethyl-2,4,5-trimethylcyclohexene B. 2-ethyl-1,4,5-trimethylcyclohexene C. 2-ethyl-1,4,5-trimethylcyclohex-1-ene D. 4-ethyl-1,2,5-trimethylcyclohexenearrow_forwardWhat is the IUPAC name of the branched alkane below? CH3 H3C. CH3 H3C ČH3 O a. 2,3-methylpropane O b. 2-ethyl-3-methylbutane Oc. trimethylpropane O d., 2, 2-dimethyl-3-methylbutane O e. 2,2,3-trimethylbutane ge Jump to..arrow_forwardP. CH3-CH2-CH3 Q. CH3-CH2-CH=CH-CH3 R. CH3-CH2-CH2-CH2-CH3 S. CH3-CH=CH-CH2-CH3 Which compounds belong to the same homologous series? P and Q Q and R R and S P and R Which structures show unsaturated hydrocarbons? P and Q P and R Q and S R and Sarrow_forward
- 2. Give the IUPAC name for the following compound. Draw the Lewis structure starting from left most carbon and make sure each carbon forms 4 single bonds. Usually alkyl groups as substituents are in the parentheses except one methyl group at the end of carbon chain ( CH3C(CH3)2CH(CH₂CH3)CH₂CH₂CH(CH3)CH3arrow_forward2. Classify each compound as an alcohol, aldehyde, sugar, ketone, methyl ketone, or hydrocarbon (there should only be one of each type of compound). Hexane Ethanol Benzaldehyde Cyclohexanone Acetone (Propanone) Glucose emplearrow_forward5. Give the IUPAC NAME of the following compound: H2C=CH(CH2)2CH=CH2 б. 6. Give the COMMON NAME of the following compound CH3 CH3-C-CH2-CH3 CH3arrow_forward
- • Question 1: Give IUPAC name for each compound. CH3 CH3-C-CH3 a. CH3CH₂CH₂-C-CH₂CH₂CH₂CH3 CH3 CH₂CH3 b. H-C-CH₂-CHCH₂ CH33 CH3 H CH₂-C-CH₂ CH₂CH3 C. CH₂CH₂CH₂-C-CH₂CH₂-C-CH3 H d. CH3 CHCH₂-C-CH3 H I CH3 • Question 2: Give IUPAC name for each compound. H CH₂CH₂CH3 રકમ જમarrow_forwardA a) Identify the functional group in each molecule by structure and by name b) Give the group of compounds to which the molecule belongs c) Explain how you name these compounds and write the IUPAC name below each compound. E. CH-OR CH CH CH₂-CH-CH₂-CH-CH B. Br -- CH: -- CH:- C = C - CH CH₂ - CH3 C. CH3- CH - C - CH: - CH3 11 CH₁ D, CH3- C - CH - CH - C OH CH₂ CH₂ S F. Br Cl CIarrow_forwardName each disubstituted benzene. Br b. CH;-CH3 a. с. CI `CH;-CH3 Brarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





