
Human Physiology
15th Edition
ISBN: 9781259864629
Author: Fox, Stuart Ira
Publisher: Mcgraw-hill Education,
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4, Problem 23RA
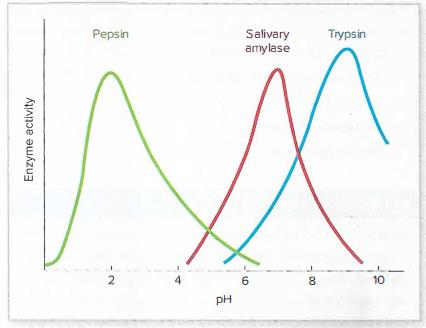
Use the graph here and in figure 4.4 to answer the following questions:
What are the pH optima of these three enzymes?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The following question focuses on how the parameters regulating enzyme function might change, and how these might appear graphically on a Michaelis-Menten plot and a Lineweaver-Burke plot. Carbonic anhydrase is an enzyme that will convert CO2 and water into HCO3.
CO2 + H20 > H+ + HCO3
There are many different isoforms of this enzyme. (see for instance
http://en.wikipedia.org/wiki/Carbonic_anhydrase
1 Assume that one variant has a Km of 10 µM and a different variant has a Km of 100 µM. Draw on the same graph a typical Michaelis-Menton plot showing the alteration in the rate of carbonic anhydrase as the CO2 level is varied for the two different variants of enzyme, assuming the concentration of the enzyme (10 mM) in the test tube is kept constant. Assume that you have equal amounts of the two different variants of carbonic anhydrase in a number of test tubes and that the Vmax for both enzymes are the same. Be sure to label the axes. For the same conditions as above, draw a…
The following question focuses on how the parameters regulating enzyme function might change, and how these might appear graphically on a Michaelis-Menten plot and a Lineweaver-Burke plot. Carbonic anhydrase is an enzyme that will convert CO2 and water into HCO3.
CO2 + H20 > H+ + HCO3
There are many different isoforms of this enzyme. (see for instance
http://en.wikipedia.org/wiki/Carbonic_anhydrase . Imidazol is a competitive inhibitor of carbonic anhydrase. It is effective at an alkaline (high) pH; in lower (more acidic) pH, it no longer inhibits the enzyme. Draw on a separate graph a Lineweaver-Burke plot for the effects of this compound at high pH and low pH. Be sure to label the axes and put in sample data points.
The following question focuses on how the parameters regulating enzyme function might change, and how these might appear graphically on a Michaelis-Menten plot and a Lineweaver-Burke plot. Carbonic anhydrase is an enzyme that will convert CO2 and water into HCO3.
CO2 + H20 > H+ + HCO3
There are many different isoforms of this enzyme. Morphine is a non-competitive inhibitor of carbonic anhydrase. Draw on the same Lineweaver-Burke plot as above a graph showing the effect of a concentration of morphine that inhibits the first enzyme such that it reduces the Vmax to ½ its maximal value. Make sure to put in sample data points. Imidazol is a competitive inhibitor of carbonic anhydrase. It is effective at an alkaline (high) pH; in lower (more acidic) pH, it no longer inhibits the enzyme. Draw on a separate graph a Lineweaver-Burke plot for the effects of this compound at high pH and low pH. Be sure to label the axes and put in sample data points.
Chapter 4 Solutions
Human Physiology
Ch. 4 - Use the lock-and-key model to explain how enzymes...Ch. 4 - Explain how enzymes are named, and the nature of...Ch. 4 - Draw graphs to represent the effects of changes in...Ch. 4 - Prob. 4CPCh. 4 - Prob. 5aCPCh. 4 - Prob. 5bCPCh. 4 - Prob. 5cCPCh. 4 - Prob. 6aCPCh. 4 - Define the terms exergonic reaction and endergonic...Ch. 4 - Prob. 7aCP
Ch. 4 - Prob. 7bCPCh. 4 - Which of these statements about enzymes is...Ch. 4 - Which of these statements about enzyme-catalyzed...Ch. 4 - Which of these statements about lactate...Ch. 4 - In a metabolic pathway, a. the product of one...Ch. 4 - In an inborn error of metabolism,
a. a genetic...Ch. 4 - Which of these represents an endergonic...Ch. 4 - Which of these statements about ATP is true? a....Ch. 4 - When oxygen is combined with 2 hydrogens to make...Ch. 4 - Enzymes increase the rate of chemical reactions...Ch. 4 - According to the law of mass action, which of...Ch. 4 - Explain the relationship between an enzyme's...Ch. 4 - Explain how the rate of enzymatic reactions may be...Ch. 4 - Explain how end-product inhibition represents a...Ch. 4 - Prob. 14RACh. 4 - The coenzymes NAD and FAD can "shuttle" hydrogens...Ch. 4 - Prob. 16RACh. 4 - Why do we need to eat food containing niacin and...Ch. 4 - Metabolic pathways can be likened to intersecting...Ch. 4 - Prob. 19RACh. 4 - Suppose you come across a bottle of enzyme tablets...Ch. 4 - Describe the energy transformations that occur...Ch. 4 - Use the reversible reactions involving the...Ch. 4 - Use the graph here and in figure 4.4 to answer the...Ch. 4 - Use the graph here and in figure 4.4 to answer the...Ch. 4 - Prob. 25RACh. 4 - Prob. 26RACh. 4 - Prob. 27RA
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- what is the Km for the following enzyme michael mentenarrow_forwardIdris has successfully extracted enzymatic proteins from the fish viscera (intestines and stomach). After homogenization and centrifugation, he managed to pool the crude enzyme extract. He is characterizing the enzymes. Please help Idris by answering the followingquestions:(a). How do I determine the enzyme activity? Please give the unit. (b) How do I get the specific activity of this enzyme? Please give the unit.arrow_forwardUsing the table below, differentiate the effect of two varying pH levels (as indicated by by the color) to the amylase enzyme. How does pH level affects the enzymatic reaction (enzyme-substrate complex)? Tube 1 Tube 2 Tube 3 Tube 4 Ingredients StarchAmylaseBuffer pH 7 StarchAmylaseBuffer pH 2 MaltoseWaterBuffer pH 7 StarchWaterBuffer pH 7 Color (1) (2) Orange Bluearrow_forward
- Table 2: Effect of pH on Enzyme Activity pH Absorbance 2 0.05 4 0.35 6 0.8 8 0.5 10 0.4 12 0.1 Use the above data table to complete the following questions: a) Plot the data “pH Vs Abs” using “connect the data point type of graph”. Label the graph with dependent and independent variables where they should be. Provide a title for the graph. b) Over what pH range does catechol oxidase catalyze catechol to benoquinone? c) Explain why the graph has a bell-shaped curve.arrow_forwardAn enzyme that catalyzes the reaction X ⇌ Y is isolated from two bacterial species. The enzymes have the same Vmax but different Km values for the substrate X. Enzyme A has a Km of 2.0 μM, and enzyme B has a Km of 0.5 μM. The plot below shows the kinetics of reactions carried out with the same concentration of each enzyme and with [X] = 1 μM. Which curve corresponds to which enzyme? Explain.arrow_forwardUse the Michaelis-Menten equation to solve the following question. If the rate of an enzymatic reaction is 5μM/min at 5 mM substrate concentration, what is the Vmax? Km is 2 mM.arrow_forward
- Rearrange the following terms to show the process of enzymatic reaction. Use and + to complete the equation. enzyme enzyme-substrate complex enzyme product substrate Several important things should be noted about this reaction: 1. A/an because of the fit between their structures; 2. As a result, something happens to the example, it might be split in two at a particular location. 3. Then the and 4. The enzyme is again. 5. Note that the arrows in the formula for enzyme reaction point acts on a specific to form a/an molecule. For comes apart, yielding the in the reaction and is now free to react _- This means that the reaction is 6. An enzyme-substrate complex can simply go back to the the 7. The products of an enzymatic reaction can react with the enzyme to form the and again; 8. It, in turn, may again form the 9. Therefore, the same. and the may act to cause a to go either way.arrow_forwardDifferentiate the concerted model and sequential model by illustrating the difference in terms of R and T forms of the enzyme when a substrate is about to bind. Write a shortdescription for each.arrow_forwardIdris has successfully extracted enzymatic proteins from the fish viscera (intestines and stomach). After homogenization and centrifugation, he managed to pool the crude enzyme extract. He is characterizing the enzymes. Please help Idris by answering the followingquestions: (a) How do I run the experiment to find the kinetic properties of the enzyme, KMand Vmax? (b) The enzyme decreased in activity in the presence of NaCl. How do I find out if NaCl is a competitive or non-competitive inhibitor? Explain.arrow_forward
- The following table provides descriptions of what is occurring at steps A, B, and C in the reaction. Review the descriptions and then choose the letter from the image that matches each description in the left-hand column of the table.arrow_forwardProduce a graph that shows the effect of pH on the rate of reaction for amylase from the tablearrow_forwardThe enzyme phosphoglucomutase catalyzes the conversion of glucose 1-phosphate to glucose 6-phosphate. After the reactants and products were mixed and allowed to reach equilibrium at 25°C, the concentration of glucose 1-phosphate was 4.5 mM and that of glucose 6-phosphate was 86 mM. Calculate Keq' and AG for this reaction. The reaction coordinate diagram for an enzyme-catalyzed reaction is shown below. How many transition states and intermediates are in the reaction? Is the reaction thermodynamically favorable? Which step is the rate-determining step of the reaction? G Reaction coordinatearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:9780134580999
Author:Elaine N. Marieb, Katja N. Hoehn
Publisher:PEARSON

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Anatomy & Physiology
Biology
ISBN:9781259398629
Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:Mcgraw Hill Education,

Molecular Biology of the Cell (Sixth Edition)
Biology
ISBN:9780815344322
Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter
Publisher:W. W. Norton & Company

Laboratory Manual For Human Anatomy & Physiology
Biology
ISBN:9781260159363
Author:Martin, Terry R., Prentice-craver, Cynthia
Publisher:McGraw-Hill Publishing Co.

Inquiry Into Life (16th Edition)
Biology
ISBN:9781260231700
Author:Sylvia S. Mader, Michael Windelspecht
Publisher:McGraw Hill Education
Enzyme Kinetics; Author: MIT OpenCourseWare;https://www.youtube.com/watch?v=FXWZr3mscUo;License: Standard Youtube License