
Hepatocellular carcinoma is the most frequent form of liver cancer. In a patient with heritable hepatocellular carcinoma, formation of the tumor was associated with eight genetic alterations affecting two different oncogenes and three different tumor-suppressor genes. These alterations are:
| i. | Mitotic recombination |
| ii. | A deletion of a chromosomal region |
| iii. | Trisomy |
| iv. | A duplication of a chromosomal region |
| v. | Uniparental disomy (see Fig. 20.24) |
| vi. | A point mutation |
| vii. | Another point mutation |
| viii. | Yet another point mutation |
For parts a–c below, supply all possible correct answers from the preceding list. Remember that the majority of point mutations are loss-of-function mutations.
| a. | Which of the mutations from the preceding list is likely to affect a proto-oncogene? |
| b. | Which of the mutations from the preceding list is likely to involve a tumor-suppressor gene? |
| c. | Which of the mutations from the preceding list involves copy-neutral loss-of-heterozygosity (that is, a loss-of-heterozygosity in which the genomes of the cancerous cells still have two copies of the gene in question, whether or not those copies are functional)? |
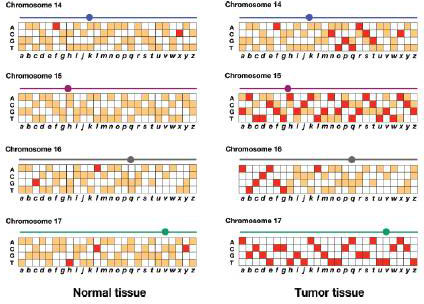
Genomic DNA is prepared from normal white blood cells and from a biopsy of the tumor in this patient. These genomic DNAs are prepared as fluorescent probes that are each hybridized to an ASO microarray of polymorphisms in the human genome (review Figs. 11.16 and 11.17). The results for SNPs a–z on chromosomes 14, 15, 16, and 17 are shown in the accompanying figure. Red and orange represent different levels of fluorescence.
| d. | Based on the microarray data, provide the most accurate localization of the first five types of genetic alterations in the list (i–v). For example, if an alteration involves markers a–e of chromosome 15, write 15a–e. |
| e. | As precisely as possible, indicate the location of the mitotic recombination event involved in the genesis of this cancer |
| f. | If these data allow you to map any of the three cancer-promoting point mutations, provide the most accurate mutation location(s) possible. |
| g. | Of all the genetic alterations i–viii, for which one do you see clear-cut evidence that the mutation or other event was inherited from a parent of the patient? |
| h. | For a tumor-suppressor gene to play a role in cancer, normally both of the copies in the tumor cells must be nonfunctional. For each of the three tumor-suppressor genes contributing to the cancer in this patient, provide a scenario explaining which two hits (i–viii in the list, with vi–viii equivalent) could be responsible, the order in which the hits must have occurred, and whether the hits in question could be inherited or could have occurred somatically. |
Want to see the full answer?
Check out a sample textbook solution
Chapter 20 Solutions
Genetics: From Genes to Genomes
- The propensity to develop retinal cancer can run in pedigrees of certain families. You have learned about the role of the retinoblastoma (Rb) protein in the cell cycle. A heterozygous individual (genotype = Rbm / Rb+) that inherits a mutant allele of the Rb gene (Rbm) as well as a normal allele of the Rb gene (Rb+) is at significant risk for developing cancer of the retina. The best explanation for this is that… A. the Rb(m) allele is dominant to the Rb(+) allele. B. a mutation in the Rb(+) allele can result in a Rb(m) / Rb(m) genotype in certain cells. C. the retina is frequently exposed to UV rays D. the Rb(m) allele directly converts the Rb(+) allele to another Rb(m) allele. E. the cells of the retina carry more than two copies of the Rb genearrow_forwardPart 3C: Below is data from a pedigree trace of patients affected with mouse model of pancreatic cancer, and below are the observations regarding a new gene called 318 that is found on chromosome 5. Patient II-4 has two copies of loss of function variant alleles (or is homozygous for loss-of-function variant 318 allele). Patient I-2 has one copy of loss of function variant and one copy of unaffected allele (is heterozygous for loss-of-function variant 318 allele). Patient II-1 has two copies of unaffected allele (or is homozygous for unaffected 318 allele). |-1 1-2 Il-1 Il-2 Il-3 |l-4 II-5 II-1 III-2 III-3 Based upon data above, what is the mode of inheritance for mouse model for pancreatic cancer? Why?arrow_forwardWhich of the following mutations will result in cancer? a. homozygous recessive mutation in a tumor-suppressor gene coding for a nonfunctional protein b. dominant mutation in a tumor-suppressor gene in which the normal protein product is overexpressed c. homozygous recessive mutation in which there is a deletion in the coding region of a proto-oncogene, leaving it nonfunctional d. dominant mutation in a proto-oncogene in which the normal protein product is overexpressedarrow_forward
- Some cancers are consistently associated with the deletion of a particularpart of a chromosome. Does the deleted region contain an oncogene or atumor-suppressor gene? Explain.arrow_forwardRetinoblastoma can be seen as a familial cancer, inherited in an autosomal recessive manner (RB-/RB-), individuals heterozygous for the RB+ and RB- alleles can develop tumor as a result of… A mitotic crossover that leads to homozygosity for RB+ in some cells and RB- in other cells A meiotic mutation in the RB+ allele that leads to homozygosity for RB+ A somatic mutation in the RB- allele that leads to homozygosity for RB+ The fact that RB- is dominant to RB+arrow_forwardGenetic tests that detect mutations in the BRCA1 and BRCA2 oncogenes are widely available. These tests reveal a number of mutations in these genes—mutations that have been linked to familial breast cancer. Assume that a young woman in a suspected breast cancer family takes the BRCA1 and BRCA2 genetic tests and receives negative results. That is, she does not test positive for the mutant alleles of BRCA1 or BRCA2. Can she consider herself free of risk for breast cancer?arrow_forward
- Acquired mutation in the p53 gene is the most common genetic alteration found in human cancer (> 50% of all cancers). A germline mutation in p53 is the causative lesion of Li- Fraumeni familial cancer syndrome. In many tumors, one p53 allele on chromosome 17p is deleted and the other is mutated. What type of protein is encoded by the p53 gene? (A) Caspase (B) DNA repair enzyme (C) Membrane cell adhesion molecule (D) Serine phosphatase (E) Telomerase (F) Transcription factor (G) Tyrosine kinasearrow_forwardAnswer the questions below: 1. Rarely , both sister chromatids of a replicated chromosome end up in one daughter cell. How might this happen and what could be the consequences of such mitotic error?2. Liver cells proliferate excessively both in patients with chronic alcoholism and in patients withliver cancer. Is alcohol an anti-apoptopic compound?arrow_forwardNeurofibromatosis 1 is considered an autosomal dominant disorder because the gene is located on the long arm of chromosome 17. It is caused by microdeletion at the long arm of chromosome 17 band 11 sub-band 2 involving the NF1 gene. Group of answer choices Statement 1 is correct. Statement 2 is incorrect Statement 1 is incorrect, statement 2 is correct Both statements are incorrect Both statements are correctarrow_forward
- A person with cancer walks into the classroom. As a biology class, we ask if we can examine some of the cancerous cells and find that many of these cancerous cells have an extra chromosome 10 (trisomy) leading to more cyclin for forming the MPF being made, leading to uncontrollable cell division (i.e. tumor and progression to cancer). What would cause extra chromosomes in cells (be it somatic or gametic cells)? Select one: a. None of these choices are linked to how sister chromatids separate and would lead to extra chromosomes in daughter cells of cell division. b. APC not functioning correctly to add ubiquitin to the cohesins, thus sister chromatids do not split correctly during cell division c. MPF itself would cause more chromosome replication d. APC is overly functioning adding cohesins to the sister chromatidsarrow_forwardIn terms of the cell cycle, what would happen if there was a defect in the Rb protein that made it unable to bind to gene regulatory proteins? (eg. the cell cycle would arrest in late G2 phase)arrow_forward4). p53 (sometimes called TP53 for “tumor protein 53") is a human tumor suppressor gene that is mutated in the majority of human cancers (many tumor types). a. For each of the mutations described below (i-iv): is this a mutation you would expect to find when sequencing p53 alleles from tumor cells? Why or why not? i. A missense mutation encoding a hyperactive form of the protein. ii. A deletion of the gene. iii. An insertion in the promoter that increases transcription 10-fold. iv. A nonsense mutation. b. When sequencing the p53 gene in tumor cells, would you expect to find only mutant version(s) of the gene or a mix of mutant and wild type versions? c. For any of the mutations you said you would expect to find in tumor cells, would you expect tumor cells to be homozygous (same mutation on both chromosomes)? Why or why not? d. Individuals with Li-Fraumeni syndrome have a very high risk of tumors originating in various tissues due to inheritance of a loss-of-function mutant allele of…arrow_forward
 Human Heredity: Principles and Issues (MindTap Co...BiologyISBN:9781305251052Author:Michael CummingsPublisher:Cengage Learning
Human Heredity: Principles and Issues (MindTap Co...BiologyISBN:9781305251052Author:Michael CummingsPublisher:Cengage Learning
