
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
4th Edition
ISBN: 9781119776741
Author: Klein
Publisher: WILEY CONS
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 6.11, Problem 18ATS
Interpretation Introduction
Interpretation: In 1994, George Olah was awarded Nobel Prize for the research on carbocation. The rearrangement of carbocation he studied occurs with the migration of the methylene group. The curved arrow is to be interpreted for the migration of the methylene group in the given transformation.
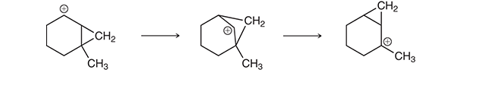
Concept introduction: In a
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
5A
In the following reactions, mixtures of alkenes and ethyl ethers
are formed. Draw their structures. Explain which is or are likely
to be the main product(s) in each reaction. In case of formation
of two isomers of alkenes, explain which is formed in greater
proportion
CH3
ofi
H3C-
-Br
CH3
EtOH
N,N-diethyl-m-toluamide (DEET) is the active ingredient in many insect repellent preparations. Following is one of the steps in its synthesis. In the box below draw the
structure of the product of this reaction.
H3C
MgBr
1. CO2
2. H3O+
product
• You do not have to consider stereochemistry.
• You do not have to explicitly draw H atoms.
• Do not include lone pairs in your answer. They will not be considered in the grading.
• Draw the Grignard reagent as a covalent magnesium bromide.
90-87
0
+
11
?
n [
10. Consider the hydrogenation reaction of compound X to Y:
H₂, Pt
X (C8H14)
→Y (C8H16)
25°C
One can conclude that X has:
A) no rings and no double bonds.
B) no rings and one double bond.
C) one ring and one double bond.
D) two rings and no double bonds.
E) one triple bond.
Chapter 6 Solutions
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
Ch. 6.1 - Prob. 1LTSCh. 6.1 - Prob. 1PTSCh. 6.1 - Prob. 2ATSCh. 6.2 - Prob. 3CCCh. 6.3 - Prob. 4CCCh. 6.3 - Prob. 5CCCh. 6.4 - Prob. 6CCCh. 6.6 - Prob. 7CCCh. 6.7 - Prob. 2LTSCh. 6.7 - Prob. 8PTS
Ch. 6.7 - Prob. 9PTSCh. 6.7 - Prob. 10ATSCh. 6.8 - Prob. 3LTSCh. 6.8 - Prob. 11PTSCh. 6.8 - Prob. 12ATSCh. 6.9 - Prob. 4LTSCh. 6.9 - Prob. 13PTSCh. 6.9 - Prob. 14ATSCh. 6.10 - Prob. 5LTSCh. 6.10 - Prob. 15PTSCh. 6.10 - Prob. 16ATSCh. 6.11 - Prob. 6LTSCh. 6.11 - Prob. 17PTSCh. 6.11 - Prob. 18ATSCh. 6 - Prob. 19PPCh. 6 - Prob. 20PPCh. 6 - Prob. 21PPCh. 6 - Prob. 22PPCh. 6 - Prob. 24PPCh. 6 - Prob. 25PPCh. 6 - Prob. 26PPCh. 6 - Prob. 27PPCh. 6 - Prob. 28PPCh. 6 - Prob. 29PPCh. 6 - Prob. 30PPCh. 6 - Prob. 31PPCh. 6 - Prob. 32PPCh. 6 - Prob. 33PPCh. 6 - Prob. 34PPCh. 6 - Prob. 35PPCh. 6 - Prob. 36PPCh. 6 - Prob. 37PPCh. 6 - Prob. 38PPCh. 6 - Prob. 39PPCh. 6 - Prob. 40PPCh. 6 - Prob. 41PPCh. 6 - Prob. 43ASPCh. 6 - Prob. 44ASPCh. 6 - Prob. 45ASPCh. 6 - Prob. 46ASPCh. 6 - Prob. 47ASPCh. 6 - Prob. 48ASPCh. 6 - Prob. 49ASPCh. 6 - Prob. 50IPCh. 6 - Prob. 51IPCh. 6 - Prob. 52IPCh. 6 - Prob. 53IPCh. 6 - Prob. 54IPCh. 6 - Prob. 55IPCh. 6 - Prob. 56IPCh. 6 - Prob. 57IPCh. 6 - Prob. 58IPCh. 6 - Prob. 59IPCh. 6 - Prob. 60IPCh. 6 - Prob. 61IPCh. 6 - Prob. 62CPCh. 6 - Prob. 64CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Alcohols undergo an oxidation reaction to yield carbonyl compounds on treatment with CrO3. For example, 2-tert-butylcyclohexanol gives 2-tert-butylcyclohexanone. If axial OH groups are generally more reactive than their equatorial isomers, which do you think reacts faster, the cis isomer of 2-tert-butylcyclohexanol or the trans isomer? Explain.arrow_forward10. A compound X of molecular formula C8H12 with no triple bonds reacts with one equivalent of H2 to give a new compound having molecular formula CaH14. What can be inferred about the structure of compound X? A) Compound X has 3 rings. B) Compound X has 3 pi bonds. Compound X has 1 ring and 2 pi bonds. D) Compound X has 2 rings and 1 pi bond. 11. Determine the product of the following reaction. H, Lindlar's cat. %3D IV A)I B) II C) III D) IV Type here to search 互i | A C 三Darrow_forward6A1: Distinguish between synthetic and retrosynthetic transformations to solve problems and predict starting materials and products. Draw any retrosynthetic sequence where a hydroxyl cycloalkane retron can be retrosynthesized into an appropriate cycloalkene, where the cycloalkene can be retrosynthesized into an appropriate cycloalkane halide. While you are allowed to work out this problem synthetically, you MUST report your final response in the retrosynthetic direction using the symbology we learned in lecture/arrow_forward
- Propene reacts with hydrogen bromide (HBr) at room temperature to produce, 1-bromopropane and 2-bromopropane. H2C=CHCH3 + HBr → CH2(Br)CH2CH3 + CH3CH(Br)CH3 Draw out the reaction mechanism of the reaction between propene and hydrogen bromide to produce 2-bromopropane. Use curly arrows to show the movement of electrons. The description should be detailed and must include the type of bond fission that takes place. You may sketch and insert suitable diagrams to aid your description if you wish. Referring to the above mechanism, explain why two products are formed in the reaction between propene and hydrogen bromide?arrow_forwardShown below is a carbocation intermediate in an electrophilic addition reaction of HCl with an alkene. For the conformation shown, select each hydrogen whose bond to carbon is aligned for effective hyperconjugation with the vacant p orbital on the positively charged carbon. Each adjacent carbon will have only one C-H bond so aligned. • Gray = C; white = H; red = 0; blue = N; dark green = Cl; brown =Br; light green =F; purple = I; yellow = S; orange = P. • Double click to select atoms. • You can zoom in and out using the mouse scroll wheel (or pinch to zoom on touch screens).arrow_forwardThe reaction shown below is carried out in two stages. In the first, the two reactants are combined in tetrahydrofuran (THF) solvent to form a neutral adduct. Aqueous acid is then added to hydrolyze this initial adduct, giving the final product. N(CH3)2 I. H a For the reaction shown above, draw the major organic product having at least one carbonyl group. • You do not have to consider stereochemistry. Pis 85 1. THF 2. H3O+ n [ ]#arrow_forward
- Acyl transfer (nucleophilic substitution at carbonyl) reactions proceed in two stages via a "tetrahedral intermediate." Draw the tetrahedral intermediate as it is first formed in the following reaction. CI H₂N OH • You do not have to consider stereochemistry. • Include all valence lone pairs in your answer. • Do not include counter-ions, e.g., Na+, I, in your answer. • In cases where there is more than one answer, just draw one. Sn [Farrow_forwardPredict the products of the following biochemical reaction: CH₂ CH CH₂ O- O || O || -(CH₂)6 - CH3 -(CH₂)6 - CH3 -(CH₂)6 - CH3 + 3 NaOH A In particular, draw the structure of the product or products P in the drawing area below. If there are no products, because this reaction won't happen, check the No reaction box under the drawing area. Note: if there is more than one product, you can draw them in any arrangement you like. Also, just draw the structure of each product. You don't have to draw the complete right-hand side of the equation, including stoichiometric coefficients. Click and drag to start drawing a structure. X :0 Śarrow_forward9. Hydrates, in which two -OH groups are connected to the same carbon, are not favorable except in a few cases. One case is: a) acetaldehyde c) acetone b) formaldehyde d) chloroform 10. The Wolff-Kishner reaction, in which an aldehyde or ketone is treated with NH2NH2 and KOH, is: a) an oxidation c) an SN2 reaction b) a reduction d) a hydration 11. Which of these is most likely to dissolve in 5% NaOH? a) 1-decanol b) decanal c) decanoic acid d) 2-decanone bu itcolf Draw tbearrow_forward
- Draw the bond-line (skeletal) structure of the compound with molecular formula C₂H₁3Br that gives the following alkene as the exclusive product of E2 elimination. Click and drag to start drawing a structure. C X C Ś c+arrow_forwardN,N-diethyl-m-toluamide (DEET) is the active ingredient in many insect repellent preparations. Following is one of the steps in its synthesis. In the box below draw the structure of the product of this reaction. H3C MgBr 1. CO₂ 2. H₂O* product • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • Do not include lone pairs in your answer. They will not be considered in the grading. • Draw the Grignard reagent as a covalent magnesium bromide.arrow_forward(dehydrobromination) A chemist carried out an elimination reaction of 1,1-dimethyl-2- bromocyclopentane. The chemist expected the reaction to yield alkene Z as product. However, alkene Z DID NOT form. Instead, three alkenes were produced: one alkene was the major product, the other two alkenes was the minor products. What was the major alkene product that formed? Br Select one: A. I B. II C. IV D. III alcohol HEAT || Z = + (HBr) IVarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY