
Figure 5.35 (left) shows the free energy curves at one particular temperature for a two-component system that has three possible solid phases (crystal structures), one of essentially pure A, one of essentially pure B, and one of intermediate composition. Draw tangent Lines to determine which phases are present at which values of x. To determine qualitatively what happens at other temperatures, you can simply shift the liquid free energy curve up or down (since the entropy of the liquid is larger than that of any solid). Do so, and construct
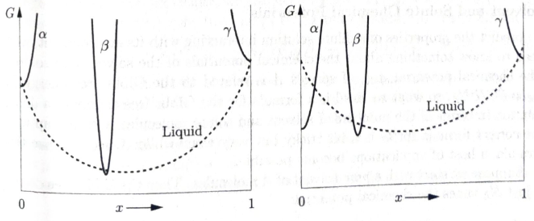
Figure 5.35. Free energy diagrams for Problems 5.71 and 5.72. a qualitative phase diagram for this system. You should find two eutectic points. Examples of systems with this behavior include water + ethylene glycol and tin + magnesium.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
An Introduction to Thermal Physics
Additional Science Textbook Solutions
Physics for Scientists and Engineers with Modern Physics
Glencoe Physical Science 2012 Student Edition (Glencoe Science) (McGraw-Hill Education)
Applied Physics (11th Edition)
Physics for Scientists and Engineers: A Strategic Approach with Modern Physics (4th Edition)
Lecture- Tutorials for Introductory Astronomy
Conceptual Integrated Science
- define the chemical potential in terms of derivatives of the ĈE energy E and enthalpy H. For a one component system, these are u= ON and Evaluate these expressions for an ideal gas and compare to ON µ = -kT In (kT/PA³ ) from H=| P,s OF and |= 1 TP ON v.rarrow_forward1 cal = 4.184 J, specific heat of iron = 0.45 J/g°C 1. If 2087.5 kcal of chemical energy is converted to mechanical energy and then electrical energy, how many kcal and joules of electrical energy is available? Explain. 2. a) Define the term, state function. ( one of the wo assmptions made for this model, (6 (amiog o) sbam od anoizulonoo ntw bas bovoado o Jominoqo brohadut eom3 sdinoesC b) Give THREE examples of state functions. (arrow_forwardAn ideal monatomic gas is contained in a cylinder with a movable piston so that the gas can do work on the outside world, and heat can be added or removed as necessary. (Figure 1) shows various paths that the gas might take in expanding from an initial state whose pressure, volume, and temperature are po, Vo, and To respectively. The gas expands to a state with final volume 4V. For some answers it will be convenient to generalize your results by using the variable R₂ = Vfinal/Vinitial, which is the ratio of final to initial volumes (equal to 4 for the expansions shown in the figure.) The figure shows several possible paths of the system in the pV plane. Although there are an infinite number of paths possible, several of those shown are special because one of their state variables remains constant during the expansion. These have the following names: Adiabatic: No heat is added or removed during the expansion. • Isobaric: The pressure remains constant during the expansion. ● •…arrow_forward
- An ideal monatomic gas is contained in a cylinder with a movable piston so that the gas can do work on the outside world, and heat can be added or removed as necessary. (Figure 1) shows various paths that the gas might take in expanding from an initial state whose pressure, volume, and temperature are po, Vo, and To respectively. The gas expands to a state with final volume 4V. For some answers it will be convenient to generalize your results by using the variable R₂ = Vfinal/Vinitial, which is the ratio of final to initial volumes (equal to 4 for the expansions shown in the figure.) The figure shows several possible paths of the system in the pV plane. Although there are an infinite number of paths possible, several of those shown are special because one of their state variables remains constant during the expansion. These have the following names: Adiabatic: No heat is added or removed during the expansion. ● Isobaric: The pressure remains constant during the expansion. ● •…arrow_forwardYour calculator can't handle enormous exponents, but we can make sense of large powers of e by converting them to large powers of 10. If we write e = 10°, then eß = (10ª)³ = 108. What is the value of a? α = 0.43 Submit ✓ Correct Part B Previous Answers What is the multiplicity of a macrostate with entropy S = 0.80 J/K? Give your answer as a power of 10. IVE ΑΣΦ = 10; aß= 1.575 1023 Submit Previous Answers Request Answer X Incorrect; Try Again Provide Feedback ?arrow_forwardProvide an equation of state (relating pressure, volume and temperature) for a real gas and interpret the terms that take into account the differences between a real and ideal gas. Comment on the extreme limits (example:zero temperature). Under what conditions does the behavior of real gases conform to the behavior expected to ideal gases?arrow_forward
- The following data (figure) describe the diffusion of a substitutional impurity atom in a polycrystalline solid. Each data set below (A, B, and C) is associated with a characteristic diffusion environment for the impurity atom. The possible diffusion environments are: surface diffusion, volume diffusion, grain boundary diffusion. Based on your understanding of solid-state diffusion mechanisms and processes, name the diffusion environment most likely associated with each data set:arrow_forwardSuppose we are dealing with an ideal gas contained in a cylinder equipped with a movable collection. The gas undergoes different types of thermodynamic transformations while interacting with the environment. Let us consider that the gas undergoes a series of transformations under different conditions of pressure and volume. For all exercises, use the general gas law PV=nRT to perform the calculations and remember to indicate the units correctly. Exercise 1 - Isothermal Transformation:During an isothermal transformation of an ideal gas, where the temperature T is held constant, the initial pressure of the gas is approximately 1250 Pa, and its final pressure is approximately 630 Pa. For ideal gases R is 0.082 atm.L/mol. K, and the temperature T is 300 K, calculate the initial and final volumes (Vi and Vf) at which the gas is during this isothermal transformation.Remember that 1 atm=101325 Pa Please make it typeable, as handwriting hinders understanding.arrow_forwardPart A: Consider a monatomic gas of particles each with mass m. What is Vx,rms =,v²), the root mean square (rms) of the x component of velocity of the gas particles if the gas is at an absolute temperature T? Express your answer in terms of T, KB, m, and other given quantities. Hint: For this case, the Equipartition Theorem reduces to m(v)=kgT. Vx,rms=J{v%) = ? Part B: Now consider the same system a monatomic gas of particles of mass m, except in three dimensions. Find vx,rms, the rms speed if the gas is at an absolute temperature T. Express your answer in terms of T, KB, m, and other given quantities.arrow_forward
- Suppose we are dealing with an ideal gas contained in a cylinder equipped with a movable collection. The gas undergoes different types of thermodynamic transformations while interacting with the environment. Let us consider that the gas undergoes a series of transformations under different conditions of pressure and volume. For all exercises, use the general gas law PV=nRT to perform the calculations and remember to indicate the units correctly. Isometric Transformation:At a pressure of 3 atm, the gas is held in a volume of 24.6 liters. It is compressed so that its temperature decreases from 400 K to 350 K. Calculate the final pressure of the gas after this isometric compression. Please make it typeable, as handwriting hinders understanding.arrow_forwardQuestion 3: In a microcanonical ensemble for 'N' non interacting molecules of an ideal gas in a specific volume 'V', how can you develop an expression for entropy (S) and change in entropy of the system if the given system obeys the Boltzmann's statistics. Using the derived value of entropy formulate an expression for ideal gas equationarrow_forwardProblem 1 The fundamental equation of thermodynamics for a system is given by G = NRT--In(T)+ In(o) where G is Gibbs free energy, N is the number of moles of chemical substance, R is gas constant, T temperature, In is the natural log function, P is pressure. Please derive an expression each for the following: is a. Entropy s as a function of temperature T, pressure p, and number of moles N. b. Volume V as a function of temperature T, pressure p, and number of moles N. c. Helmholtz free energy as a function of temperature T, pressure p, and number of moles N.arrow_forward
 College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON
University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press
Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley
Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON
College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON





