
Figure 5.32 shows the phase diagram of plagioclase feldspar, which can be considered a mixture of albite
- Suppose you discover a rock in which each plagioclase crystal varies in composition from center to edge, with the centers of the largest crystals composed of 70% anorthite and the outermost parts of all crystal made of essentially pure albite. Explain in some details how this variation might arise. What was the composition of the liquid magma from which the rock formed?
- Suppose you discover another rock body in which the crystals near the top are albite–rich while the crystals near the bottom are anorthite–rich. Explain how this variationmight arise.
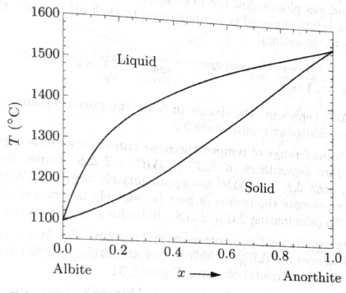
Figure 5.32. The phase diagram of plagioclase feldspar (at atmospheric pressure). From N. L. Bowen, “The Melting Phenomena of the Plagioclase Feldspars,” American Journal of Science 35, 577–599 (1913).
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
An Introduction to Thermal Physics
Additional Science Textbook Solutions
Applied Physics (11th Edition)
Sears And Zemansky's University Physics With Modern Physics
College Physics: A Strategic Approach (4th Edition)
Physics: Principles with Applications
The Cosmic Perspective Fundamentals (2nd Edition)
- Graphite has hexagonal crystal lattice which contains four atoms per unit cell at positions of (0 0 0), (1/3 2/3 0), (O 0 1/2) and (2/3 1/3 1 12). Show that the structure factor is given by the following equations. Hint: e^ix+e^- .ix=2cosxarrow_forwardUsing drawing and Explanation. P-Si Show (Draw) the 2D atomic crystal structure of p type silicon. Do Both fixed and mobile charges should be clearly observable.arrow_forwardFluids were sampled from surface manifestations within a geothermal system. Laboratory analysesresulted to the following concentrations: Silica quartz = 500 ppm Amorphous silica = 1000 ppm Sodium = 1000 ppm Potassium = 200 ppm Magnesium = 0.2 ppm Tabulate the possible temperatures of the geothermal system according to each geothermometer (inorder as presented). What do you think is the valid temperature range of the area? present yor solutions.arrow_forward
- Would you expect a silicon wafer doped with antimony to be a p-type or n-type wafer? Why? What about a silicon wafer doped with gallium? Please answer in typing format please yarrow_forwardThe edge length of a unit cell of a crystal is 4.3 Angstrom and an atomic mass of 90 g/mol. If the density of the crystal is 9.02 g/cm3, find the number of atoms in a unit cell.arrow_forwardA crystalline solid is made up of two elements ‘A’ and ‘B’. Atoms of A are present at thecorners and atoms of B are present at face centers. One atom A is missing from the corner.Find the simplest formula of the solid.arrow_forward
- The element neptunium has a density of 19.6 g/ml. What is the radius of a sphere of this material if it has a mass of 70kg? V=4/3pi r^3arrow_forwardplease help me out. details and explanations are very much appreciated.arrow_forwardOrthoclase feldspar is a mineral that commonly occurs in certain intrusive igneous rocks that crystallize from melts deep within the crust of the earth. The chemical formula for orthoclase is KAISİ3O3 and its molecular weight is 278.4 g/mol. Most potassium in orthoclase is 39K (93.2581%). However, .0117% of naturally occuring potassium is 40K (which is unstable with a half- life of 1.28x109 yr). When orthoclase-bearing igneous rocks form, the ortho- clase crystallizes and its chemical constituents are locked in the crystal structure, no longer able to be freely exchanged with their environment. As this happens, the 40K in the orthoclase continues to decay according to 40K → 40Ar + e* + Ve The decay produces 40Ar, but the argon remains trapped in the rigid crystal structure. As time passes, argon accumulates in the crystal structure. Many years after the crystallization of the igneous rock, a crystal of ortho- clase from the rock may be extracted so that its argon content can be…arrow_forward
- 100 Mll, N 8.6.E (a) Calculate the equilibrium temperature of a meteoroid of mass M, density p, and albedo A in the vicinity of the Earth. (b) Evaluate your result for a chondrite with M = 10° g, p = 2.5 g cm-3, and albedo A = 0.05 and for an achondrite with M = 10° g, p = 3 g cm and albedo A = 0.3. -3arrow_forwardQ- Explain all characteristics of 7 crystal classes and 14 crystal types with their neet diagrams.arrow_forwardA 10 cm long core sample with a 1 inch (2.54 cm) diameter is sampled from a reservoir. Unfortunately, during transportation some portion of the sample is cracked and lost. Now the sample is not a perfect cylinder and it is not possible to cut a cylinder sample from the rest of the sample. The core sample is cleaned for porosity analysis. The mass of the core sample is measured as 100 g. The core sample is placed in cell two of a Boyle's Law device (Helium porosimeter). Each of the cells has a volume of 200 cc. Cell one is pressured to 100.0 psig. Cell two is evacuated. Then, the cells are connected and the resulting pressure is 48 psig. The sample is clean sandstone composed of silica and the density of silica is 2.65 g/cm³. Calculate porosity.arrow_forward
 College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON
University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press
Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley
Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON
College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON





