
Concept explainers
(a)
Interpretation: The type of structures needs to be determined.

Concept Introduction:Structural isomers have the same chemical formula, but the atoms arrangements are different in both structures.
(b)
Interpretation: The type of structures needs to be determined.
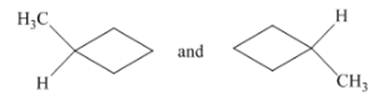
Concept Introduction:The identical structures have same groups, but different positions.
(c)
Interpretation: The type of structures needs to be determined.
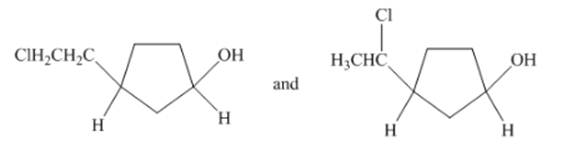
Concept Introduction:Structural isomers have same molecular formula, but the arrangements of atoms and groups are different.
(d)
Interpretation: The type of structures needs to be determined.
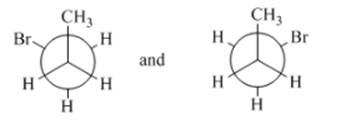
Concept Introduction:Conformers have the same formula, but the isomers can be interchanged by rotation at an angle.
(e)
Interpretation: The type of structures needs to be determined.
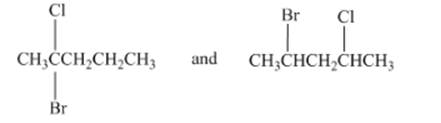
Concept Introduction:Structural isomers have same formula but different arrangements of molecules and atoms.
(f)
Interpretation: The type of structures needs to be determined.

Concept Introduction:Stereoisomer has same molecular formula, but the arrangements of the groups are different in 3-D space.
(f)
Interpretation: The type of structures needs to be determined.

Concept Introduction:Stereoisomer has same molecular formula, but the arrangements of the groups are different in 3-D space.
(g)
Interpretation: The type of structures needs to be determined.
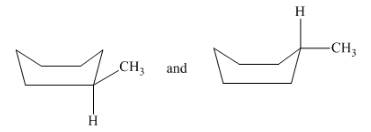
Concept Introduction:Conformers have the same formula, but the groups are arranged in a different manner.
(h)
Interpretation: The type of structures needs to be determined.
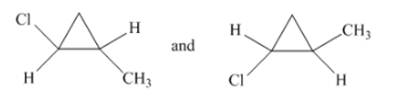
Concept Introduction:Stereoisomer has same molecular formula, but the position of the groups is different in 3-D space.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Organic Chemistry: Structure and Function
- Distinguish between isomerism and resonance. Distinguish between structural and geometric isomerism. When writing the various structural isomers, the most difficult task is identifying which are different isomers and which are identical to a previously written structurethat is, which are compounds that differ only by the rotation of a carbon single bond. How do you distinguish between structural isomers and those that are identical? Alkenes and cycloalkanes are structural isomers of each other. Give an example of each using C4H8. Another common feature of alkenes and cycloalkanes is that both have restricted rotation about one or more bonds in the compound, so both can exhibit cis- trans isomerism. What is required for an alkene or cycloalkane to exhibit cis-trans isomerism? Explain the difference between cis and trans isomers. Alcohols and ethers are structural isomers of each other, as are aldehydes and ketones. Give an example of each to illustrate. Which functional group in Table 21-4 can be structural isomers of carboxylic acids? What is optical isomerism? What do you look for to determine whether an organic compound exhibits optical isomerism? 1-Bromo-1-chloroethane is optically active whereas 1-bromo-2-chloroethane is not optically active. Explain.arrow_forwardCompounds X and Y both have the formula C7H14. Both X and Y react with one molar equivalent of hydrogen in the presence of a palladium catalyst to form 2-methylhexane. The heat of hydrogenation of X is greater than that of Y. Both X and Y react with HCI to give the same single C₂H₁5Cl compound as the major product. What is the structure of X? • In cases where there is more than one answer, just draw one. 7 0▾ ChemDoodleⓇ 146arrow_forwardExplain What are conformational isomerism?arrow_forward
- Butanal (C4H&O) has eight H atoms. Suppose that any of these H atoms can be replaced by a Cl atom to yield a molecule with the formula C4H¬CIO. (a) Identify two H atoms where this substitution would yield constitutional isomers of C4H7CIO; (b) enantiomers of C4H;CIO; (c) conformers of C4H;CIO. H. H H H нн H Butanal O=Uarrow_forwardDraw all possible isomers of trimethylcyclopropane. Name them. Which of these isomers are chiral? Indicate by underlining the name(s).arrow_forwardWhich of the following conformers has the highest energy (the least stable)?Justify your choicearrow_forward
- Which of the following statements are correct? * Conformation exist in a dynamic equilibrium state. Conformers differ largely in energies. Conformers cannot be separated. Amount of more stable conformers is more than that of less stable conformers at equilibrium.arrow_forwardWhich of the following is an incorrect statement: Isomers have the same molecular formula such as cyclohexane and 1-hexene. Conformational isomers are enantiomers or diastereomers, which can interconvert through rotations around bonds within molecules, which include anti and gauche butane. Stereoisomers have the same connectivity but different arrangement of their parts in space, which include enantiomers, diastereomer, and conformational isomers, which include cis and trans dichlorocyclobutane. O Structural or constitutional isomers are molecules of the same formula but different connectivity such as 1,1-dichlorocyclopropane and 1.2-dichlorocyclobutane.arrow_forwardCompound W shows stereoisomerism. Draw spatial formulae to show this isomerism Describe the physical property which could be used to distinguish between the Isomers.arrow_forward
- Using your knowledge of (and the definition of) cis and trans isomers and the following fact(s) about Cis/Trans Isomers please answer the following: (FACT(S): With multiple double bonds in a compound the TRANS-TRANS configuration is the most stable conformation. With multiple double bonds in a compound the CIS-CIS configuration is the least stable conformation.) Which structure is the least stable based on the information given?arrow_forwardS.3. Show in each of the pairs below whether the two structures are structural isomers, stereoisomers, or different Conformations of the same compound.arrow_forwardThe meiting points and boiling points of two isomeric alkanes are as follows: CH3(CH2);CH3, mp = -57 °C and bp = 126 °C; (CHalsCC(CH3)3, mp = 102 °C and bp = 106 °C. (a) Explain why one isomer has a lower melting point but higher boiling point. (b) Explain why there is a small difference in the boiling points of the two compounds, but a huge difference in their melting points.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning



