
(a)
Interpretation:
The exergonic portion present in given reaction should be identified.
Concept Introduction:
Exergonic: The reaction is considered as exergonic if energy released since the reactants loses its energy, making the free energy is more negative hence making it spontaneous reaction.
Endergonic: The reaction is considered as endergonic if it needs more energy means that activation energy is much higher making the reaction non spontaneous.
(a)
Answer to Problem 21.20UKC
The exergonic portion is conversion of succinyl phosphate into succinate.
Explanation of Solution
Analyzing the given reaction it is clear that conversion of succinyl phosphate to succinate is exergonic since the phosphate bond in succinyl phosphate gets cleaved which releases energy.
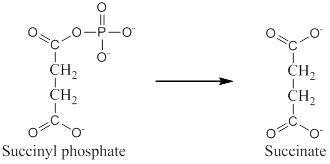
Hence, exergonic portion is conversion of succinyl phosphate into succinate.
(b)
Interpretation:
The endergonic portion present in given reaction should be identified.
Concept Introduction:
Exergonic: The reaction is considered as exergonic if energy released since the reactants loses its energy making the free energy more negative hence making it spontaneous reaction.
Endergonic: The reaction is considered as endergonic if it needs more energy means that activation energy is much higher making the reaction non spontaneous.
(b)
Answer to Problem 21.20UKC
The endergonic portion is conversion of
Explanation of Solution
Analyzing the given reaction it is clear that conversion of
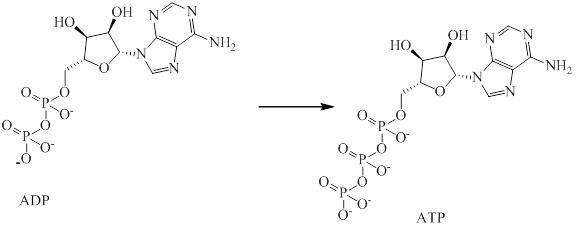
Therefore, addition of phosphate group into
Hence, endergonic portion is conversion of
Want to see more full solutions like this?
Chapter 21 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- Which letter represents the ΔG of the reaction?arrow_forwardDraw the product AND propose a reasonable, detailed stepwise mechanism, using curved arrow notation to show the flow of electrons, for the following reaction.arrow_forwardUnder standard conditions, will the following reaction proceed spontaneously as written?arrow_forward
- In the preparation of aspirin, You can do the functional group test to ensure the completion of the reaction. (a) What is the name of reagent used? (b) What is your observation if any unreacted starting material is present? (c) What is the name of the functional group responsible for this reaction?arrow_forwarddiscuss the following statement: “Whether the ΔG for a reaction is larger, smaller, or the same as ΔG° depends on the concentration of the compounds that participate in the reaction.”arrow_forwardIdentify the major and minor product(s) of the following reaction:arrow_forward
- Why are catabolic reactions considered exergonic and anabolic reactions considered endergonic?arrow_forwardif the reaction given below occurs and pure A and B were mixed, which of the following would take place as equilibrium was established A + B ⇌ C a. the concentration of C would increase for a time, then remain constant b. the concentration of A would increase for a time, then decrease c. the concentration of B would increase for a time, then remain constantarrow_forwardIf you measured the rate of reaction at 20°C to be 1.11 x 10-5 M/s when using 0.080 M I1- and 0.040 M S2O82-. Approximately how long will the reaction take if you were to increase the temperature to 30 °C?arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





