
Concept explainers
(a)
Interpretation:
Structure of
Concept Introduction:
Amides are synthesized using amidification reaction. This involves a reaction between
(a)
Answer to Problem 17.134EP
Carboxylic acid that is required was,
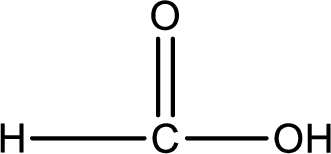
Explanation of Solution
Given structure of compound is,
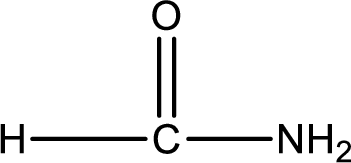
As the nitrogen atom present in the above amide has two hydrogen atoms bonded to it, the amide is a primary amide. Primary amide is produced by the reaction of ammonia with carboxylic acid. The “parent” carboxylic acid can be identified as shown below,
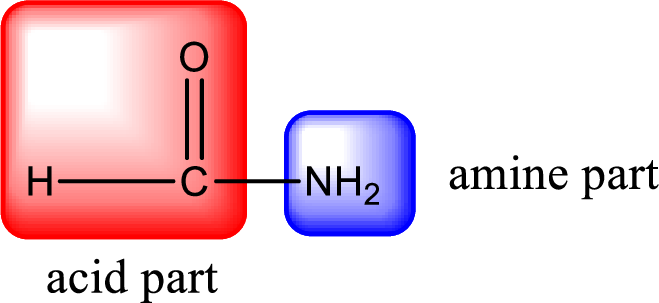
Hydrogen atom has to be added to the amine part and
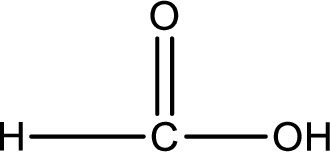
Structure of carboxylic acid that is required to produce the given compound is drawn.
(b)
Interpretation:
Structure of carboxylic acid that is required to produce the given compound as product through amidification has to be given.
Concept Introduction:
Amides are synthesized using amidification reaction. This involves a reaction between amine and carboxylic acid. In this reaction, the
(b)
Answer to Problem 17.134EP
Carboxylic acid that is required was,
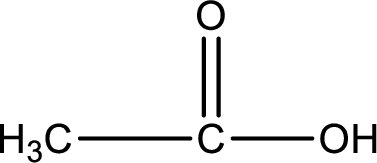
Explanation of Solution
Given structure of compound is,
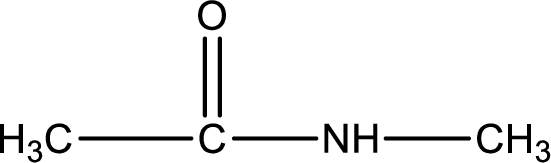
As the nitrogen atom present in the above amide has two hydrogen atoms bonded to it, the amide is a primary amide. Primary amide is produced by the reaction of ammonia with carboxylic acid. The “parent” carboxylic acid can be identified as shown below,
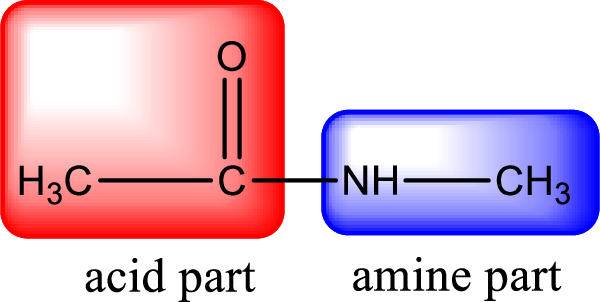
Hydrogen atom has to be added to the amine part and
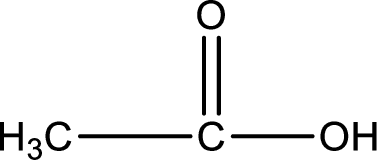
Structure of carboxylic acid that is required to produce the given compound is drawn.
(c)
Interpretation:
Structure of carboxylic acid that is required to produce the given compound as product through amidification has to be given.
Concept Introduction:
Amides are synthesized using amidification reaction. This involves a reaction between amine and carboxylic acid. In this reaction, the
(c)
Answer to Problem 17.134EP
Carboxylic acid that is required was,
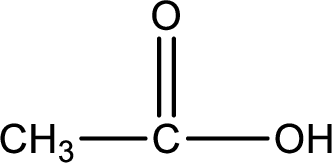
Explanation of Solution
Given structure of compound is,
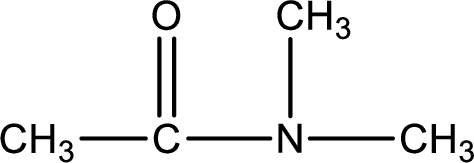
As the nitrogen atom present in the above amide has two hydrogen atoms bonded to it, the amide is a primary amide. Primary amide is produced by the reaction of ammonia with carboxylic acid. The “parent” carboxylic acid can be identified as shown below,
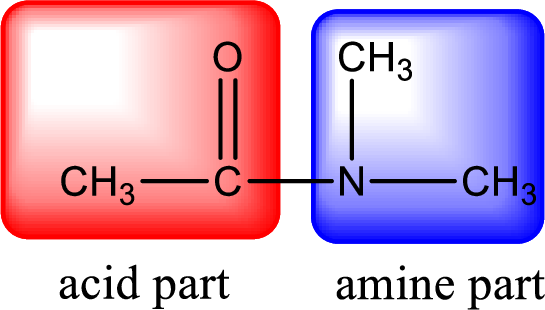
Hydrogen atom has to be added to the amine part and
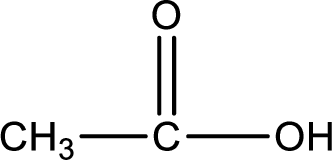
Structure of carboxylic acid that is required to produce the given compound is drawn.
(d)
Interpretation:
Structure of carboxylic acid that is required to produce the given compound as product through amidification has to be given.
Concept Introduction:
Amides are synthesized using amidification reaction. This involves a reaction between amine and carboxylic acid. In this reaction, the
(d)
Answer to Problem 17.134EP
Carboxylic acid that is required was,
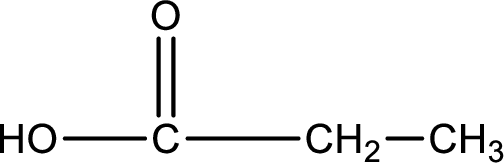
Explanation of Solution
Given structure of compound is,
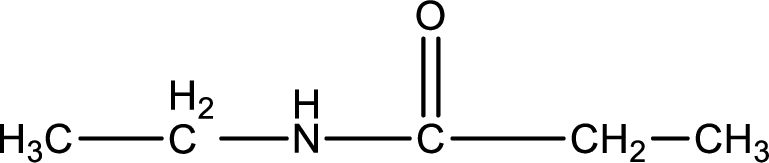
As the nitrogen atom present in the above amide has two hydrogen atoms bonded to it, the amide is a primary amide. Primary amide is produced by the reaction of ammonia with carboxylic acid. The “parent” carboxylic acid can be identified as shown below,
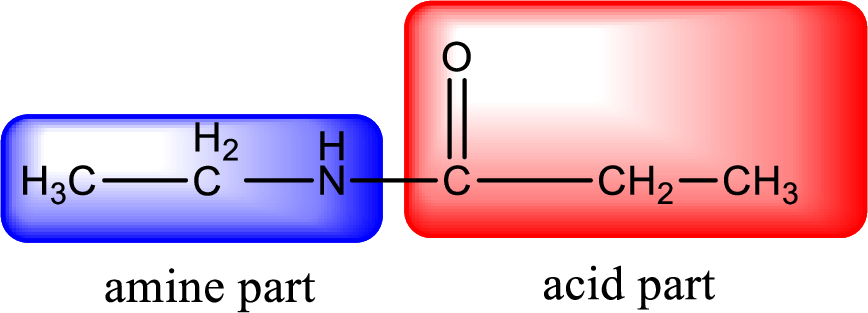
Hydrogen atom has to be added to the amine part and
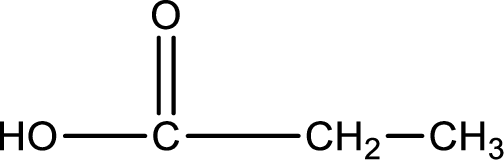
Structure of carboxylic acid that is required to produce the given compound is drawn.
Want to see more full solutions like this?
Chapter 17 Solutions
General, Organic, and Biological Chemistry
- The Ksp for lead iodide ( Pbl₂) is 1.4 × 10-8. Calculate the solubility of lead iodide in each of the following. a. water Solubility = mol/L b. 0.17 M Pb(NO3)2 Solubility = c. 0.017 M NaI mol/L Solubility = mol/Larrow_forwardPleasssssseeee solve this question in cheeemsirty, thankss sirarrow_forwardPleasssssseeee solve this question in cheeemsirty, thankss sirarrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning



