
(a)
Interpretation:
Structure of amine that is formed when the given amine salt reacts with has to be drawn.
Concept Introduction:
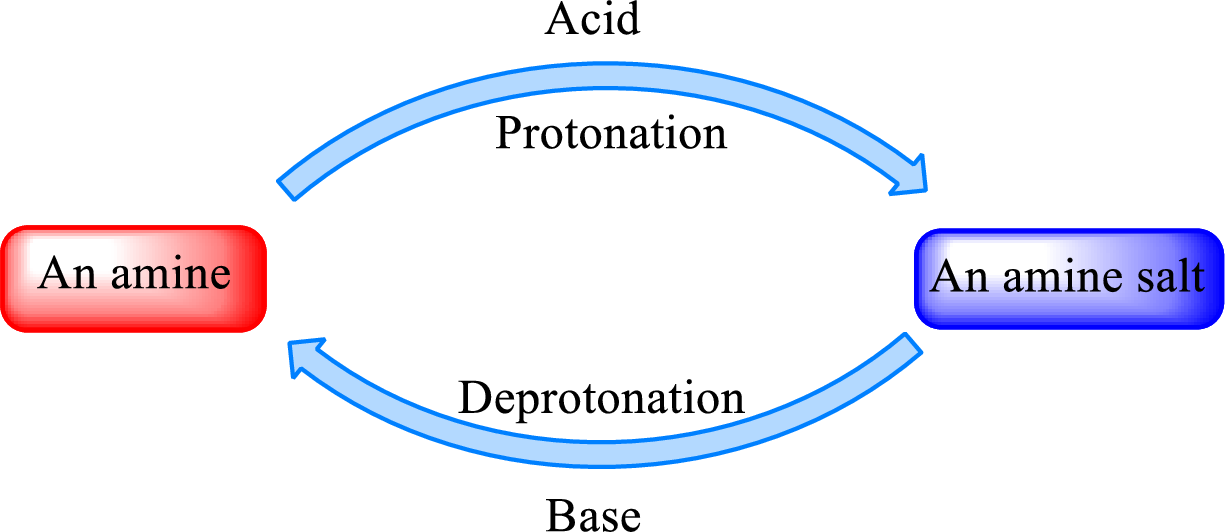
Neutralization reaction is the one that takes place between an acid and a base to give salt as product. As
When a strong base is added to the amine salt, the parent amine can be obtained. This is a reverse reaction of the amine salt formation reaction. These reactions can be represented as shown below,
(b)
Interpretation:
Structure of amine that is formed when the given amine salt reacts with has to be drawn.
Concept Introduction:
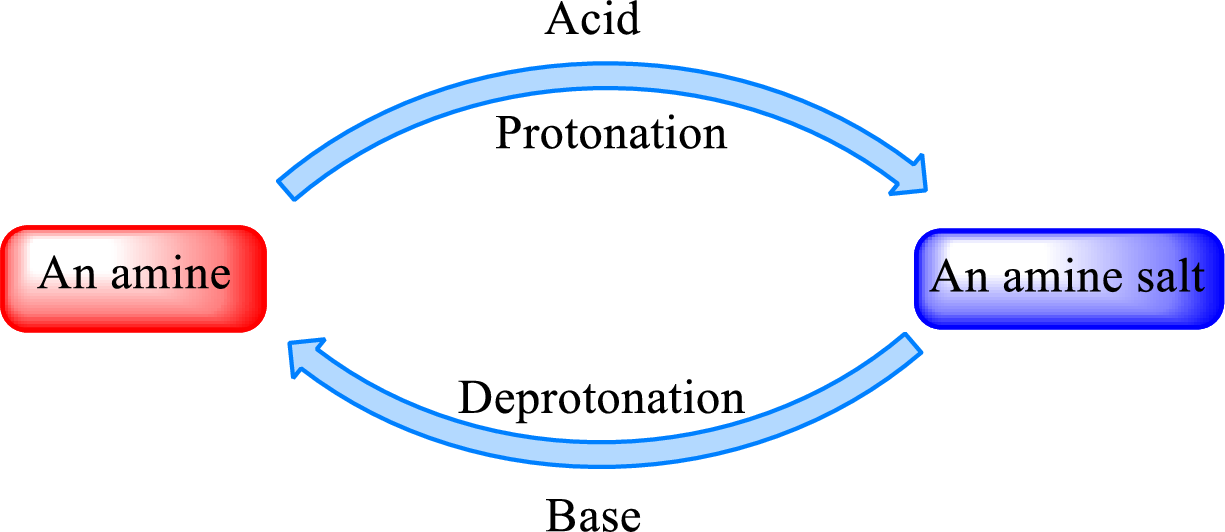
Neutralization reaction is the one that takes place between an acid and a base to give salt as product. As amines are bases due to the amino group in it, the reaction with inorganic acid or carboxylic acid gives salt as product. The salt formed is an amine salt. Proton is donated from the acid to the nitrogen atom which acts as a proton acceptor. In simple words, it can be said that in an amine‑acid reaction, the acid loses a hydrogen ion and amine gains a hydrogen ion.
When a strong base is added to the amine salt, the parent amine can be obtained. This is a reverse reaction of the amine salt formation reaction. These reactions can be represented as shown below,
(c)
Interpretation:
Structure of amine that is formed when the given amine salt reacts with has to be drawn.
Concept Introduction:
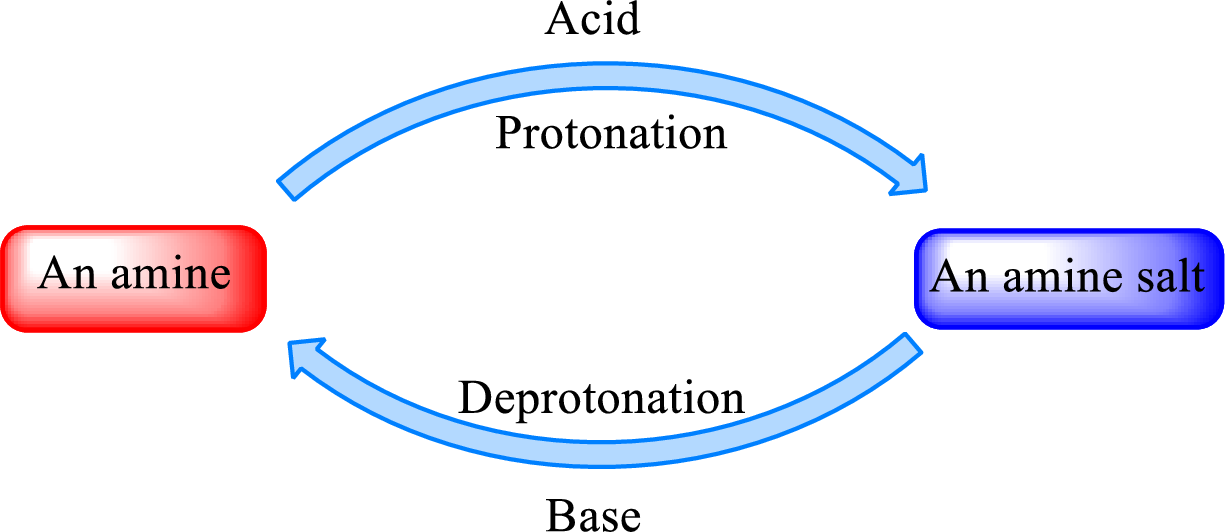
Neutralization reaction is the one that takes place between an acid and a base to give salt as product. As amines are bases due to the amino group in it, the reaction with inorganic acid or carboxylic acid gives salt as product. The salt formed is an amine salt. Proton is donated from the acid to the nitrogen atom which acts as a proton acceptor. In simple words, it can be said that in an amine‑acid reaction, the acid loses a hydrogen ion and amine gains a hydrogen ion.
When a strong base is added to the amine salt, the parent amine can be obtained. This is a reverse reaction of the amine salt formation reaction. These reactions can be represented as shown below,
(d)
Interpretation:
Structure of amine that is formed when the given amine salt reacts with has to be drawn.
Concept Introduction:
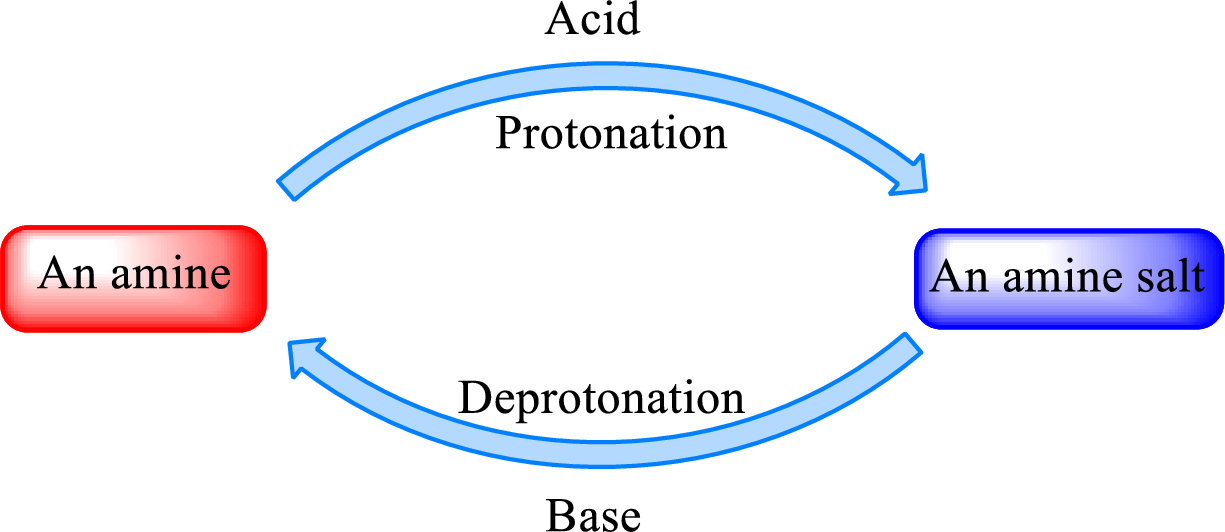
Neutralization reaction is the one that takes place between an acid and a base to give salt as product. As amines are bases due to the amino group in it, the reaction with inorganic acid or carboxylic acid gives salt as product. The salt formed is an amine salt. Proton is donated from the acid to the nitrogen atom which acts as a proton acceptor. In simple words, it can be said that in an amine‑acid reaction, the acid loses a hydrogen ion and amine gains a hydrogen ion.
When a strong base is added to the amine salt, the parent amine can be obtained. This is a reverse reaction of the amine salt formation reaction. These reactions can be represented as shown below,
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
General, Organic, and Biological Chemistry
- Q1 - What type(s) of bonding would be expected for each of the following materials: solid xenon, calcium fluoride (CaF2), bronze, cadmium telluride (CdTe), rubber, and tungsten? Material solid xenon CaF2 bronze CdTe rubber tungsten Type(s) of bonding Q2- If the atomic radius of lead is 0.175 nm, calculate the volume of its unit cell in cubic meters.arrow_forwardDetermine the atomic packing factor of quartz, knowing that the number of Si atoms per cm3 is 2.66·1022 and that the atomic radii of silicon and oxygen are 0.038 and 0.117 nm.arrow_forwardUse the following data for an unknown gas at 300 K to determine the molecular mass of the gas.arrow_forward
- 2. Provide a complete retrosynthetic analysis and a complete forward synthetic scheme to make the following target molecule from the given starting material. You may use any other reagents necessary. Brarrow_forward146. Use the following data for NH3(g) at 273 K to determine B2p (T) at 273 K. P (bar) 0.10 0.20 0.30 0.40 0.50 0.60 (Z -1)/10-4 1.519 3.038 4.557 6.071 7.583 9.002 0.70 10.551arrow_forward110. Compare the pressures given by (a) the ideal gas law, (b) the van der Waals equation, and (c) the Redlic-Kwong equation for propane at 400 K and p = 10.62 mol dm³. The van der Waals parameters for propane are a = 9.3919 dm6 bar mol-2 and b = 0.090494 dm³ mol−1. The Redlich-Kwong parameters are A = 183.02 dm bar mol-2 and B = 0.062723 dm³ mol-1. The experimental value is 400 bar.arrow_forward
- Research in surface science is carried out using stainless steel ultra-high vacuum chambers with pressures as low as 10-12 torr. How many molecules are there in a 1.00 cm3 volume at this pressure and at a temperature of 300 K? For comparison, calculate the number of molecules in a 1.00 cm3 volume at atmospheric pressure and room temperature. In outer space the pressure is approximately 1.3 x 10-11 Pa and the temperature is approximately 2.7 K (determined using the blackbody radiation of the universe). How many molecules would you expect find in 1.00 cm3 of outer space?arrow_forwardDraw the predominant form of arginine at pH = 7.9. The pKa of the side chain is 12.5. Include proper stereochemistry. H2N OH NH H₂N 'N' છ H pH = 7.9 Select to Drawarrow_forwardPlease correct answer and don't used hand raitingarrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning









