
Concept explainers
(a)
Interpretation: The structure of the product formed from acid-catalyzed hydration of
Concept introduction: Acid-catalyzed hydration is the electrophilic addition of water. The reactive species that act as a catalyst is
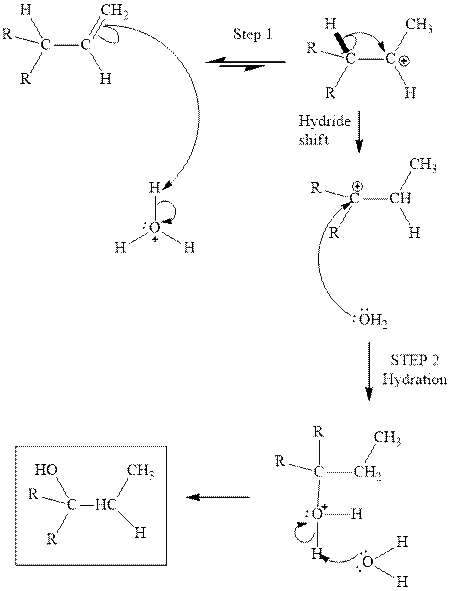
(b)
Interpretation: The structures of isomeric alcohols with chemical formula
Concept introduction: Acid-catalyzed hydration is the electrophilic addition of water. The reactive species that act as a catalyst is
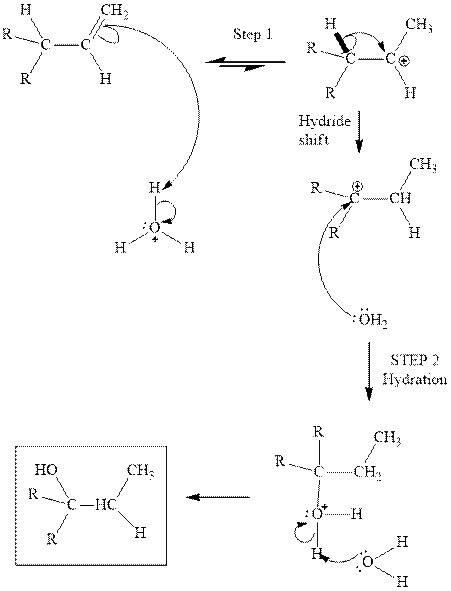
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
- When 2-pentene is treated with Cl2 in methanol, three products are formed. Account for the formation of each product (you need not explain their relative percentages).arrow_forwardA compound X of molecular formula C7H14 when hydrogenated produces 2,4-dimethylpentane and when hydroborated the obtained alcohol is 2,4-dimethyl-1-pentanol. a. What is the structure of X? b. If X is reacted with H2O catalyzed by H2SO4, what product is obtained? Write the structure, give the name, and write the complete mechanism, step by step of this reaction, in such a way as to explain the formation of the compound (DO NOT FORGET THE CORRECT USE OF ARROWS!).arrow_forwardYou are required to synthesize 2-bromopentane from the reaction between an alkene with HBr. Which alkene, 1-pentene or 2-pentene, should you react with HBr in order to get 2-bromopentane? Give an explanation.arrow_forward
- 12.Use of KMnO, to effect oxidative cleave of the alkene J, C3H16, yields two fragments, one of which is butanoic acid and the other a ketone, Q. When J reacts with one molar equivalent of H-Cl, the alkyl halide K, C3H17CI, is formed. What are the structures of J, J, and Q? Write all the reactions, and show your reasoning.arrow_forwardAlkenes undergo an addition reaction with borane in tetrahydrofuran (THF). For the reaction below: BH3 THF (solvent) a Draw the structure of the major organic product. Use the wedge/hash bond tools to indicate stereochemistry where it exists. Use wedge and hash bonds ONLY when needed to show reaction stereochemistry. If the reaction produces a racemic mixture, just draw one stereoisomer. opy aste ChemDoodlearrow_forwardOzonolysis (O3 in CH2Cl2) of compound A under reducing conditions (Zn /acetic acid) gives formaldehyde, 2-butanone, and compound B. Catalytic hydrogenation (H2/Pd) of A gives 2,7-dimethylnonane. What is a possible structure for compound A? a. 2,7-Dimethyl-2,8-nonadieneb. 2,7-Dimethyl-1,8-nonadienec. 2,7-Dimethyl-1,6-nonadiened. 2,7-Dimethyl-1,7-nonadienearrow_forward
- An alkene having the molecular formula C,H14 is treated sequentially with ozone (O3) and zinc/acetic acid to give the product/s shown. Draw a structural formula for the alkene. • You do not have to consider stereochemistry. You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one. opy astearrow_forwardМЕСНANISMS 1,2-epoxy-1-methylcyclopentane undergoes both acid-catalyzed and base-catalyzed opening of the epoxide ring to form two different products. > Using ethanol as the solvent and an appropriate acid, show the steps in the acid catalyzed mechanism, writing structures for all products of the steps. Circle the major product(s). > Using ethanol as the solvent and an appropriate base, show the steps in the base- catalyzed mechanism, writing structures for all products of the steps. Circle the major product(s). 1,2-epoxy-1-methylcyclopentane CH3arrow_forwardGive the condensed structure and the IUPAC name of the product/s for each of the following reactions and indicate for each the mechanism involved in the formation of such produc/s as SN1, SN2, E1, E2 a. CH;CH;CH;Br + LIAIH, b. (CH3),CBr + CH; CH;OH, heat at 60 C c. BrCH;CH2CH,Br + Mg(ether)arrow_forward
- An alkene having the molecular formula C5H₁0 is treated sequentially with ozone (03) and zinc/acetic acid to give the product/s shown. H3C CH3 ● H3C Draw a structural formula for the alkene. • You do not have to consider stereochemistry. You do not have to explicitly draw H atoms. In cases where there is more than one answer, just draw one.arrow_forward0 CH₂ → ?? If the above compound is subjected to combustion, what are the products? ethanoic acid +2-phenyl-ethene-1-ol 15CO₂ (g) + 5H₂0 (g) O 2-phenyl-ethyl-ethanoate + H₂O 20CO₂ (g) + 10H₂O(g) →?? CH, if the above compound was treated with H and H₂O what product will form?arrow_forward4. Draw structural formulas for all possible monohalogenation products that might be formed in the following reactions, then assign them IUPAC names. (a) + Cl₂ lightarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
