
Interpretation:The reason behind the prohibition of mineral oil usage for melting point determination of
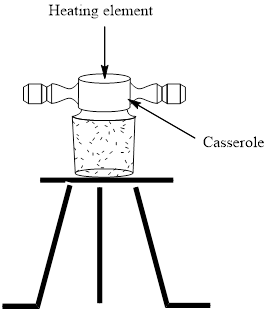
Concept introduction:In order to monitor the temperature while melting point is recorded the temperature of the bath is checked by several heating devices such as oil baths that are electrically heated. They can be brought to a certain temperature by insertion of Nichrome resistance wire in the bath. A magnetic stirrer may be kept within the oil bath to ensure uniform temperature by thermal equilibration. The basic oil bath has a heating element that can be either heated over Bunsen burner or electrically with Nichrome wire.
Trending nowThis is a popular solution!

Chapter 10 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
- Explain the principle behind the operation of Clinistix® Reagent Strips.arrow_forwardwhat would the chemical reaction be for an experiment completing esterification of combining acetic acid, 1-butanol, which i believe gives n-butyl acetate as reaction. What would theoretical yield then be/how is it calculated?arrow_forwardProvide a brief justification for the following procedures in drug production. 1.Storing magnesium citrate in the refrigerator. 2. Filtering solution while its hot. 3. Cooled purified water in preparing calcium hydroxide solution. 4. Sterilization of materials for syrup preparation. 5. Addition of potassium iodide in Lugol’s solution.arrow_forward
- what is the role of methanol for extracting chlorophyll? What molecular force holds your chlorophyll and solvent together?arrow_forward105 100 95 75 0 1 2 3 4 5 67 8 9 10 11 12 13 14 15 16 17 18 19 20 Volume distillate collected (mL) 1. Indicate the volume of the distillate at which you should stop collecting to make sure that the distillate is only composed of one component. 2. Indicate the boiling point range involved in the collection of this amount of distillate. Temp in Still Head ("C)arrow_forward1. A student wants to dry a solution of aniline in hexane. Which drying agent should he use for the task? Rationalize the choice. 2. If calcium oxide is used as a drying agent, what is obtained as a product? 3. Why is ill-advised to use P4O10 as drying agents for “wet” acetone?arrow_forward
- give at least three characteristics of dichloromethane that make it a good extracting solvent for alkaloidarrow_forwardWhy do banana and apple show discoloration when it left out in the open air for some time?arrow_forwardHow long the time I need to dryied the g-C3N4 under vacuum at 60 C after washed and filtered?arrow_forward
- 1. What type of reaction explains the solubility of phenol in Sodium Hydroxide? Explain the organic product formed 2. How do the reactions of phenol samples with FeCl3 compare? Which structural component of the phenols account for the observation? 3. What compound is the precipitate formed in the Bromine water test? 4. Write the reaction formed in the formation of phenolphthalein. Identify the functional group in phenolphthalein , which is responsible for the indicator property. 5. What is the significance of Millon's test?arrow_forward1. What is the reason behind the portion-by-portion evaporation of DCM in the extraction of eugenol? 2. Enumerate some pharmaceutical and medical uses of eugenol 3. What does the ferric chloride test identify? Is it specific test for eugenol? Why or why not?arrow_forwardGive a brief chemical structural analysis of Sildenafil citrate (viagra)with active functional groups. Reference if possible.arrow_forward
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
