
Concept explainers
(a)
Interpretation: Structure of the major product expected from the ionic addition of
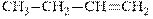
Concept introduction:Alkenes are considered electron-rich and undergo bromination addition reaction with
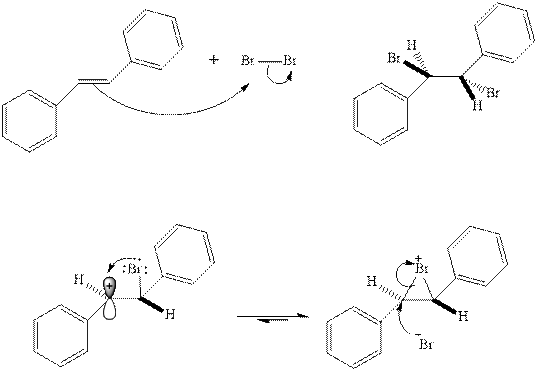
The mechanism of
In the second step, the strained bromonium ion intermediate opens while
(b)
Interpretation: Structure of the major product expected from the ionic addition of
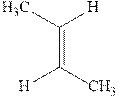
Concept introduction:Alkenes are considered electron-rich and undergo bromination addition reaction with
The mechanism of
In the second step, the strained bromonium ion intermediate opens while
(c)
Interpretation: Structure of the major product expected from the ionic addition of
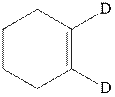
Concept introduction:Alkenes are considered electron-rich and undergo bromination addition reaction with
The mechanism of
In the second step, the strained bromonium ion intermediate opens while
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
- Draw the structural formula for the alkene with molecular formula C5H10 that will react to give the following productsarrow_forwardWhat are the products formed from the following reactions: 1) The addition of dimethylborane (Me2BH) to ethylene 2) The addition of dimethylborane (Me2BH) to acetylene Please include the formulas for the products as well. Thank you!arrow_forward∆H° values obtained for a series of similar reactions are one set of experimental data used to determine the relative stability of alkenes. Explain how the following data suggest that cis-but-2-ene is more stable than but-1-ene (Section 12.3A).arrow_forward
- Which alcohols can be prepared as a single product by hydroboration– oxidation of an alkene? Which alcohols can be prepared as a single product by the acid-catalyzed addition of H2O to an alkene?arrow_forwarda) What products would you expect from the elimination reaction of 3-Bromo-2- methylpentane? Show the reaction by writing the condensed structural formula of the reactants and products. Identify the major and minor products. b) What alkyl halide might the 3,6-Dimethyl-1- heptene have been made from?arrow_forwarddraw the two possible carbocations that can form when this alkene reacts with a strong acid (such as HBr or H3O+). of the two structures you drew, circle the more stable carbocationarrow_forward
- A hydrocarbon of unknown structure with the formula CgH14 was isolated. In an effort to determine the structure, the compound was subjected to hydrogenation with H2 and Pd, which afforded a product with a formula of C8H16 Which of the following compounds are possible candidates for the starting hydrocarbon?@GMU 2020 Me II II Mearrow_forwardWhat is the molecular formula of the following alkene? CI o CH14CI o CH13CI o CH15CI o CH11CIarrow_forwardWhich products are formed when hydrobromic acid is added to (a) trans-2-hexene, (b) 2-methyl- 2-pentene, and (c) 4-methylcyclohexene, and how many regioisomers can be formed in each case?arrow_forward
- 2-chloropropane is a major product of the reaction of chlorine with propane under ultraviolet light. Write the mechanism for this reaction including the initiation step and the two propagation steps.arrow_forward5 Write the molecular formula for each alkane. Y \ y t (a) (c) HE8 iw 146 (b)- eriam in Beris mal bond HO H ox dT O HOH w ar HO HO (s) HOarrow_forwardDraw the structure of the reactant and name the product of the following reactions: 3-methyl-2-heptanone + NaBH4arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning


