
Interpretation:The mistake made in procedure that might result in appearance of unusual red-orange solid should be identified.
Concept introduction:Alkenes are considered electron-rich and undergo bromination addition reaction with
The mechanism of
In the second step, the strained bromonium ion intermediate opens while
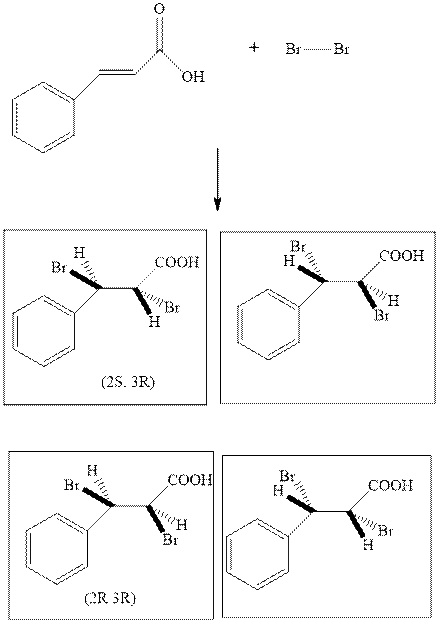
The bromination of
yields four stereoisomers illustrated as follows:
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
- 7) Explain why the diastereoselectivity is so much better in the first reaction versus the second one. Ph- Ph- m-СРВА + errintioner 95:5 anti:syn Ph- Ph- m-CPBA + enantiomer 61:39 canti:synarrow_forward3) Complete the following: Stereochemistry and regiochemistry may be important. b) d) type type MeO Br type type type OH H Br E1 Reaction Borohydride Reduction H NaOAc DMF Асон HO PCC CH₂Cl2 H CO₂Et f) g) h) i) j) type type type type type OH OH Hydride Reduction MeHN Reductive Amination HO Br OHarrow_forwardIdentify how IR spectroscopy might be used to monitor the progress of the following reaction. [conc. H,SO,]arrow_forward
- Pyridine exhibits a UV transition of the type n at 270 nm. In this transition, one of the unshared electrons on nitrogen is promoted from a nonbonding MO to a (antibonding) MO. What is the effect on this UV peak if pyridine is protonated?arrow_forwardHow is benzophenone suited for nueleophilie aromatic Sub Stitution which positions and the ring can be Substitutebarrow_forwardWhat is the best way to remeber the differences between Sn1, Sn2, E1 and E2 reations?arrow_forward
- 20:24 1 : 10. Draw the full cleavage of 2-ethoxypropane by HCI scheme on your paper, and then draw in the curved arrows for each step of the mechanism. HICI co OⓇ ← H₂O 11. What reagent can be used to convert 2-methylbutan-1-ol into 2-methylbutanal? A) Bleach B) LIAIH4 C) DMSO, (COCI)2, TEA D) Na₂Cr₂O7, H₂SO4 E) 03 12. Fill in the box with the major organic product of the first step of the reaction shown? H-Cl Send a chat Oarrow_forwardQUESTION 18 Consider the ether cleavage reaction shown below. What mechanism occurs and what are the major organic products of the reaction? 1 eq. HCI ponding letter designation O A. Mechanism: S2, Products: B.. Mechanism: SN1, Products: OC. Mechanism: SN1, Products: CI HO D.. Mechanism: SN2, Products: HOarrow_forwardDraw all the resonance forms for the sigma complexes corresponding to bromination ofaniline at the meta, and para positionsarrow_forward
- V. What is the mechanism of this?arrow_forwardd) Explain why product C is racemic but product D is non-racemic. Br CH,OH fonnet Siz mechinmwhin mes in inversion of the procdnct odue to Hhen bukside ata from the Mactoprile Br NaSH Darrow_forwardPlease deaw structures of the resonance structures of "enolate" of the following compounds with "a-H's" . Each has more than one kind, so choose the a-H marked (*). Thank you!arrow_forward

 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning



