
(a)
Interpretation:The mechanism for the formation of molecule 26 from molecule 28 through a hydride shift followed by deprotonation needs to be explained.
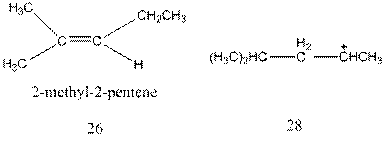
Concept Introduction : A carbocation rearrangement in which a hydrogen atom migrates from the carbon to the carbon possessing formal charge of one from the adjacent carbonis called hydride shift.
(b)
Interpretation:The mechanism for formation of 27 from 28 needs to be determined.
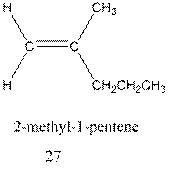
Concept Introduction : A carbocation rearrangement in which a hydrogen atom migrates from the carbon to the carbon possessing formal charge of one from the adjacent carbonis called hydride shift.
(c)
Interpretation:The mechanism of formation of 26 through acid catalysed rearrangement of either isomer of 4-methyl-2-pentene and formation of 27 by addition of a proton to 26 followed by deprotonation of intermediate carbocation needs to be explained.
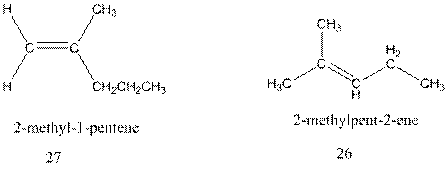
Concept Introduction : A carbocation rearrangement in which a hydrogen atom migrates from the carbon to the carbon possessing formal charge of one from the adjacent carbonis called hydride shift.
(d)
Interpretation:The different in the two types of mechanism for the formation of 26 and 27 needs to be explained.
Concept Introduction:Mechanism of a
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
- 8. Carry out the following syntheses: a. Cyclopentene to n-propylcyclopentane b. Cyclopentanol to n-propylcyclopentanearrow_forward2. Name and Draw the structures p-chloroaniline of all possible chemical (Electrophilic aronmatic substitution) reactions of the compound namely a. Halogenation (Chlorination or Bromination) b. Nitration c. Sulphonation d. Friedal Craft Alkylation e. Eriedal Craft Acylationarrow_forward26) What type of reaction is depicted below? NaCe thenH* BO a. 1,2-addition b. acyl substitution c. conjugate addition d. SN reaction e. an impossible one; carbanions do not add to alkenesarrow_forward
- 37. Under acidic condition, which of the following compounds can not be oxidized by KMnO4. A. toluene B. 2-butene C. methylcyclohexane D. 2-butyne 38. What alkene product can be produced from the reaction of 2-hexyne with Lindlar's catalyst? A. cis-2-Hexene B. trans-2-Hexene C. Hexane D. A and Barrow_forward46. Which carbonyl group of the given compound is most reactive for nucleophilic addition reaction? A. Carbonyl Group 1 B. Carbonyl Group 2 C.Carbonyl Group 3 D. All have equal reactivity 47. Which of the following does NOT give a brick red precipitate with Fehling solution? A Acetone B. Acetaldehyde C. Formalin D. D-glucose 48. Which of the following structures contains a hemiacetal group CO OH H₂C 0 OCH, A OH OCH H₂C-C-CH₂ C C.H. CH OCH OCH, barrow_forward37- The Sulphonation of benzene ring proceeds by using . ???? SO,H (A) Conc.HNO3 and conc. H2S04 (C) Conc. H2SO4 (B) Dil. H2SO4 (D) SO3/ H2SO4 38- Which of the following benzene derivative is the most reactive and ortho, para director? CHO NH. (A) (B) (C) (D) 39- What is the major product for the following reaction? NO, HNO, ???? H,SO, NO, NO, NO, NO (A) NO, (B) NO, (D) None of these 40- a – D - glucose differ from B-D-glucose (A) Because they are anomer (B) Because they are enantiomers (C) Because they are mirror image of each other (D) Because they are geometrical isomers 41-Ninhydrin test is given by (C) Proteins (D) Carbohydrates (B) Alcohols (A) Alkynes 42- Which test of the following can distinguish between 1°, 2° and 3° amines : (C) Acidic Test (D) None (B) Tolen's Test (A) The Hinsberg Test 43- How many a – amino acid are there in animal protein : (D) 10 (B) 20 (C) 15 (A) 55arrow_forward
- Show the reaction mechanism for the formation of a. RCONH2 from the reaction of RCOOR' with NH3 b. RCOOR" from the reaction of RCOOR' with R"OH in acidic mediumarrow_forwardi. LIAIH4 i. deprotection J ii. dehydration .CO2Me CH2=CHCOCH3 protection ii. H30* K The synthesis of K from H involves a 3-steps reaction as shown above. Hydrolysis of H followed by heating produces compound L. Give IUPAC name for L. Write the reaction mechanism involved during the heating process that brought iii. to the formation of L.arrow_forwarda. Use the following reaction to determine how much of each product would be releasedif 42 000 kg (42 tonnes) of methyl isocyanate reacted with excess water:methyl isocyanate + water → 1,3-dimethyl urea + carbon dioxide2C2H3NO + H2O → C3H8N2O + CO2arrow_forward
- select reagents: cyclopentanone Reagents a. C6H5CHO b. NaOH, ethanol c. Pyrrolidine, cat. Hj. d. H₂C=CHCN e. H3O+ f. LDA g. A) bg EtOC(=O)CO₂Et h. i. B) ka k. I. m. O: BrCH₂CH=CH₂ NatOEt, ethanol Br₂, H+ K+ t-BUO™ CH2(CO2Et)z heat C) jh CO₂Et D) Imarrow_forward15. Give the major organic product, paying attention to stereochemistry (syn/anti). Indicate if a racemic mixture is expected. a. a. BH3 b. H2O2, OH a. BH3 b. b. H2O2, OH a. BH3 a. BH3 C. d. b. H2O2, OH b. H₂O2, OH e. a. BH3 b. H₂O2, OHarrow_forward27. a. Label the reactive features of the following reactants, select the most reactive feature, then write and highlight what it needs. Also, state if a carbocation, carbon radical, or carbanion will start to develop, and/or if aromatic character will be lost as a result of a reaction between these molecules. hex-4-en-3-one + CH;SH b. Use mechanism arrows to illustrate the reaction that occurs. If applicable, use stabilization resources to deal with the carbocation, carbon radical, or carbanion that starts to develop during the reaction, and draw the structure of any resonance- stabilized intermediate. Continue labelling and diagramming the reaction until you find the major stable product(s). Finally, state the stereochemistry of the major product(s) and use either Fisher projection or perspective formula representations to illustrate that stereochemistry.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





