
(a)
Interpretation: The shape of the compound
Concept Introduction: VSEPR theory stands for Valence Shell Electron Pair Repulsion theory.
Scientists used the VSEPR theory to explain the three-dimensional shape of molecules.
(a)
Answer to Problem 60A
The given compound
Explanation of Solution
The given compound is
To predict the shape, draw the structure of the molecule.
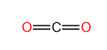
Since there are only two bonding pairs, the shape of the molecule is linear.
(b)
Interpretation: The shape of the compound
Concept Introduction: VSEPR theory stands for Valence Shell Electron Pair Repulsion theory.
Scientists used the VSEPR theory to explain the three-dimensional shape of molecules.
(b)
Answer to Problem 60A
The given compound
Explanation of Solution
The given compound is
To predict the shape, draw the structure of the molecule.
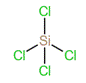
Since there are only four bonding pairs, the shape of the molecule is tetrahedral.
(c)
Interpretation: The shape of the compound
Concept Introduction: VSEPR theory stands for Valence Shell Electron Pair Repulsion theory.
Scientists used the VSEPR theory to explain the three-dimensional shape of molecules.
(c)
Answer to Problem 60A
The given compound
Explanation of Solution
The given compound is
To predict the shape, draw the structure of the molecule.
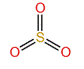
Since there are only three bonding pairs and no lone pairs, the shape of the molecule is trigonal planar.
(d)
Interpretation: The shape of the compound
Concept Introduction: VSEPR theory stands for Valence Shell Electron Pair Repulsion theory.
Scientists used the VSEPR theory to explain the three-dimensional shape of molecules.
(d)
Answer to Problem 60A
The given compound
Explanation of Solution
The given compound is
To predict the shape, draw the structure of the molecule.
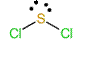
Since there are two bonding pairs and two lone pairs, the shape of the molecule is bent.
(e)
Interpretation: The shape of the compound
Concept Introduction: VSEPR theory stands for Valence Shell Electron Pair Repulsion theory.
Scientists used the VSEPR theory to explain the three-dimensional shape of molecules.
(e)
Answer to Problem 60A
The given compound
Explanation of Solution
The given compound is
To predict the shape, draw the structure of the molecule.

Since there are only one bonding pairs and one lone pairs, the shape of the molecule is linear.
(f)
Interpretation: The shape of the compound
Concept Introduction: VSEPR theory stands for Valence Shell Electron Pair Repulsion theory.
Scientists used the VSEPR theory to explain the three-dimensional shape of molecules.
(f)
Answer to Problem 60A
The given compound
Explanation of Solution
The given compound is
To predict the shape, draw the structure of the molecule.
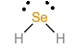
Since there are two bonding pairs and two lone pairs, the shape of the molecule is bent.
Chapter 8 Solutions
Chemistry 2012 Student Edition (hard Cover) Grade 11
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





