
Interpretation: The number of electrons shared between two atoms to form double and triple covalent bonds needs to be determined.
Concept Introduction: Covalent bonds are formed by sharing of electrons between two atoms. Here both atoms cannot gain or lose electrons to complete their octets, but they share electron pairs.
Explanation of Solution
A covalent bond is formed by sharing of electrons between two atoms. If two atoms share one pair of electrons between two atoms, a single bond is formed. In the case of a double bond, two electron pairs are shared between two atoms. For example, carbon and oxygen have double bonds between them in
molecule. This is represented as follows:
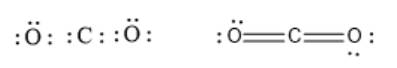
Similarly, triple bonds are formed when three electron pairs are shared between two atoms. Thus, in the case of a triple bond, 3 bonding electron pairs will be there between two atoms. For example, nitrogen atoms have a triple bond between them in
molecule. This is represented as follows:
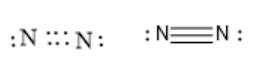
Thus, the number of electrons shared between two atoms is 4 (2 electron pairs) in the case of a double bond and 6 (3 electron pairs) in the case of a triple covalent bond.
Chapter 8 Solutions
Chemistry 2012 Student Edition (hard Cover) Grade 11
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





