
(a)
Interpretation:
The temperature dependence of the vapor pressure of phosphoryl chloride difluoride is given, ln p against 1/T has to be plotted.
Concept introduction:
The relationship to plot is In P versus 1/T is given below.
(a)
Explanation of Solution
Given,
| Temperature, T/K | 190 | 228 | 250 | 273 |
| Vapour pressure, P/Torr | 3.2 | 68 | 240 | 672 |
Unit conversion is given below,
Therefore,
| Temp. | V.P (Torr) | V.P (atm) | lnP | |
The ln p against 1/T plot is given below,
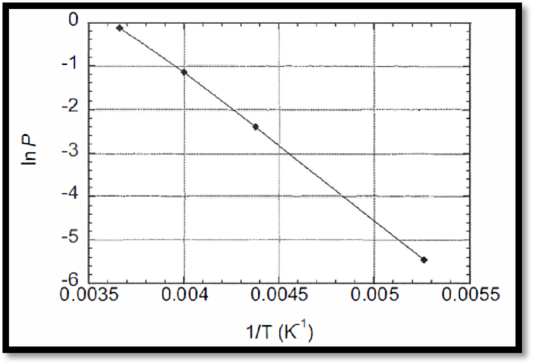
Figure 1
From the curve, slope is given below,
(b)
Interpretation:
The enthalpy of the vaporization has to be calculated.
Concept introduction:
Refer to part (a)
(b)
Explanation of Solution
The relationship to plot is In P versus 1/T, this gives a straight line.
From the curve, slope is given below,
The enthalpy of the vaporization is
(c)
Interpretation:
The entropy of the vaporization has to be calculated.
Concept introduction:
Refer to part (a)
(c)
Explanation of Solution
The relationship to plot is In P versus 1/T, this gives a straight line.
From the curve, slope is given below,
The entropy of the vaporization is
(d)
Interpretation:
The normal boiling point of phosphoryl chloride difluoride has to be calculated.
Concept introduction:
Refer to part (a)
(d)
Explanation of Solution
The normal boiling point of phosphoryl chloride difluoride is calculated when the vapor pressure is
Therefore, it will happen when.
The normal boiling point of phosphoryl chloride difluoride is
(e)
Interpretation:
If the pressure is 15 torr, temperature of phosphoryl chloride difluoride has to be calculated.
Concept introduction:
Refer to part (a)
(e)
Explanation of Solution
The temperature of phosphoryl chloride difluoride value is calculated as follows,
The temperature of phosphoryl chloride difluoride value is
Want to see more full solutions like this?
Chapter 5 Solutions
Chemical Principles: The Quest for Insight
- In a coffee-cup calorimeter, 1.60 g NH4NO3 is mixed with 75.0 g water at an initial temperature of 25.00C. After dissolution of the salt, the final temperature of the calorimeter contents is 23.34C. Assuming the solution has a heat capacity of 4.18 J/C g and assuming no heat loss to the calorimeter, calculate the enthalpy change for the dissolution of NH4NO3 in units of kJ/mol.arrow_forwardWhich of the following quantities can be taken to be independent of temperature? independent of pressure? (a) H for a reaction (b) S for a reaction (c) G for a reaction (d) S for a substancearrow_forwardCalculate the standard Gibbs free-energy change when SO3 forms from SO2 and O2 at 298 K. Why is sulfur trioxide an important substance to study? (Hint: What happens when it combines with water?)arrow_forward
- Another step in the metabolism of glucose, which occurs after the formation of glucose6-phosphate, is the conversion of fructose6-phosphate to fructose1,6-bisphosphate(bis meanstwo): Fructose6-phosphate(aq) + H2PO4(aq) fructose l,6-bisphosphate(aq) + H2O() + H+(aq) (a) This reaction has a Gibbs free energy change of +16.7 kJ/mol of fructose6-phosphate. Is it endergonic or exergonic? (b) Write the equation for the formation of 1 mol ADP fromATR for which rG = 30.5 kJ/mol. (c) Couple these two reactions to get an exergonic process;write its overall chemical equation, and calculate theGibbs free energy change.arrow_forwardDetermine the standard Gibbs free energy change, rG, for the reactions of liquid methanol, of CO(g), and ofethyne, C2H2(g), with oxygen gas to form gaseous carbondioxide and (if hydrogen is present) liquid water at298 K. Use your calculations to decide which of thesesubstances are kinetically stable and which are thermodynamically stable: CH3OH(), CO(g), C2H9(g), CO2(g),H2O().arrow_forwardUse the appropriate tables to calculate H for (a) the reaction between copper(II) oxide and carbon monoxide to give copper metal and carbon dioxide. (b) the decomposition of one mole of methyl alcohol (CH3OH) to methane and oxygen gases.arrow_forward
- Consider the reaction NH4+(aq) H+(aq)+NH3(aq) Use G f for NH3(aq) at 25C=26.7 kJ/mol and the appropriate tables to calculate (a) G at 25C (b) Ka at 25Carrow_forwardWhen one mol of KOH is neutralized by sulfuric acid, q=56 kJ. (This is called the heat of neutralization.) At 23.7C, 25.0 mL of 0.475 M H2SO4 is neutralized by 0.613 M KOH in a coffee-cup calorimeter. Assume that the specific heat of all solutions is 4.18J/gC, that the density of all solutions is 1.00 g/mL, and that volumes are additive. (a) How many mL of KOH is required to neutralize H2SO4? (b) What is the final temperature of the solution?arrow_forwardCalculate H when a 38-g sample of glucose, C6H12O6(s), burns in excess O2(g) to form CO2(g) and H2O() in a reaction at constant pressure and 298.15 K.arrow_forward
- What is the sign of the standard Gibbs free-energy change at low temperatures and at high temperatures for the explosive decomposition of TNT? Use your knowledge of TNT and the chemical equation, particularly the phases, to answer this question. (Thermodynamic data for TNT are not in Appendix G.) 2C7H5N3O6(s) 3N2(g) + 5H2O() + 7C(s) + 7CO(g)arrow_forwardDetermine the equilibrium constant for the reaction Sn+Pb2+Sn2++Pbarrow_forwardWould the amount of heat absorbed by the dissolution in Example 5.6 appear greater, lesser, or remain the same if the heat capacity of the calorimeter were taken into account? Explain your answer.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





