
Concept explainers
(a)
Interpretation:
The electron-dot structure of the given structure is to be drawn and described.
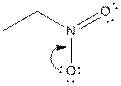
Concept introduction:
The expression to calculate the formal charge of the substance is shown below:
The electron dot structures can be described as the structure in which electrons are represented around the atoms in the molecule.
In the resonance structure, the electrons are moved towards the more electro negativity atoms and more positive charges. The arrow is originated from pi electrons or unshared electrons, and move towards the more electronegative atoms and positive charges.
(b)
Interpretation:
The electron-dot structure of the given structure is to be drawn and described.
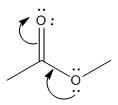
Concept introduction:
The expression to calculate the formal charge of the substance is shown below:
The electron dot structures can be described as the structure in which electrons are represented around the atoms in the molecule.
In the resonance structure, the electrons are moved towards the more electro negativity atoms and more positive charges. The arrow will be originated from pi electrons or unshared pair of electrons and move towards the more electronegative atoms and positive charges.
(c)
Interpretation:
The electron-dot structure of the given structure is to be drawn and described.
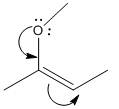
Concept introduction:
The expression to calculate the formal charge of the substance is shown below:
The electron dot structures can be described as the structure in which electrons are represented around the atoms in the molecule.
In the resonance structure, the electrons are moved towards the more electro negativity atoms and more positive charges. The arrow will be originated from pi electrons or unshared pair of electrons andmove towards the more electronegative atoms and positive charges.
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
CHEMISTRY-TEXT
- Draw the best Lewis structure(s) for the following molecular formulas:arrow_forward1. Draw the resonance structures of the following molecule, using curved arrows to show electron movement and appropriate arrows to show the relationship between the structures. All lone pairs and charges must be clearly shown.arrow_forwardDraw the Lewis structures for the following four molecules, being sure to show all steps following the methods covered in class. Structures without work shown will be marked incorrect. Also, one of these molecules has resonance structures – for this compound, make sure to include all resonance structures, indicate formal charges for each atom. SO2 OF2 IF3 NH4+arrow_forward
- complete the following lewis structure (draw preferred) and fill in the table. show formal charges on lewis structure.arrow_forwardDraw all resonance structures for the following and show the arrows for moving the electronsarrow_forwardGive typed answer not written What are resonance structures for these molecules?arrow_forward
- Determin the total number of resonance structures for the following image below?arrow_forwardIf the atom "X" is in the fifth period of the periodic table, identify the element. Be sure toclearly explain your answer. Note: Ignore the positive formal charge on the X. Please answer the question in the image. Thank you :)arrow_forwardWhich statement best describes the structure that results from the following curved arrow: ? The resulting structure is a valid resonance structure. The resulting structure is NOT a valid resonance structure, because a single bond has been broken. O The resulting structure is NOT a valid resonance structure, because the octet rule has been violated.arrow_forward
- Consider the following two resonance structures: Does the arrow pushing pattern represents the flow of electrons that converts the first resonance structure into the second resonance structure? And if not draw the correct bond line structure.arrow_forwardDraw the Lewis structure for the following compoundarrow_forwardDraw the resonance structure for the following molecule: Just started with resonance, where would the arrows go? I'm unsure of this problem.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





