
CHEMISTRY-TEXT
8th Edition
ISBN: 9780134856230
Author: Robinson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 23, Problem 23.16A
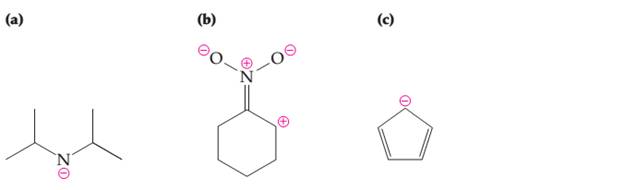
Draw complete electron-dot structures from the line drawing by adding lone pairs of electrons and hydrogen atoms.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Differentiate between double and triple Covalent Bonds?
what are the differences between ionic and covalent bond?
Draw a Lewis dot structure then from your dot structure predict the molecular shape for each of the following. Draw (using dots) all electrons on appropriate atoms.
a) IOF5(I is the central atom)
b) ClOF2 (Cl is the central atom)
Chapter 23 Solutions
CHEMISTRY-TEXT
Ch. 23 - Prob. 23.1PCh. 23 - Prob. 23.2ACh. 23 - Prob. 23.3PCh. 23 - Prob. 23.4ACh. 23 - Prob. 23.5PCh. 23 - Prob. 23.6ACh. 23 - Draw a complete electron-dot structure for...Ch. 23 - Limonene is the major component in the oil of...Ch. 23 - Draw an orbital overlap picture for methyl a mine...Ch. 23 - Prob. 23.10A
Ch. 23 - Draw an orbital overlap picture for formaldehyde,...Ch. 23 - Prob. 23.12ACh. 23 - Prob. 23.13PCh. 23 - Prob. 23.14ACh. 23 - Prob. 23.15PCh. 23 - Draw complete electron-dot structures from the...Ch. 23 - Prob. 23.17PCh. 23 - Prob. 23.18ACh. 23 - Draw a simplified orbital overlap picture of...Ch. 23 - Prob. 23.20ACh. 23 - Prob. 23.21PCh. 23 - Prob. 23.22ACh. 23 - Prob. 23.23PCh. 23 - Prob. 23.24ACh. 23 - Prob. 23.25PCh. 23 - Prob. 23.26ACh. 23 - Asparagine is a naturally occurring aminoacid that...Ch. 23 - Prob. 23.28PCh. 23 - Prob. 23.29PCh. 23 - Prob. 23.30CPCh. 23 - Prob. 23.31CPCh. 23 - Prob. 23.32CPCh. 23 - Prob. 23.33CPCh. 23 - Prob. 23.34CPCh. 23 - Prob. 23.35CPCh. 23 - The following structure is a representation of...Ch. 23 - Prob. 23.37CPCh. 23 - Prob. 23.38CPCh. 23 - Prob. 23.39CPCh. 23 - Prob. 23.40SPCh. 23 - Prob. 23.41SPCh. 23 - Draw a straight-chain alkane with 6 carbon atoms...Ch. 23 - Prob. 23.43SPCh. 23 - Prob. 23.44SPCh. 23 - Prob. 23.45SPCh. 23 - The following line drawings represent two amino...Ch. 23 - Prob. 23.47SPCh. 23 - Prob. 23.48SPCh. 23 - Prob. 23.49SPCh. 23 - Prob. 23.50SPCh. 23 - Prob. 23.51SPCh. 23 - Prob. 23.52SPCh. 23 - Prob. 23.53SPCh. 23 - Prob. 23.54SPCh. 23 - Prob. 23.55SPCh. 23 - Prob. 23.56SPCh. 23 - Prob. 23.57SPCh. 23 - Prob. 23.58SPCh. 23 - Give a line drawing and molecular formula for the...Ch. 23 - Prob. 23.60SPCh. 23 - Prob. 23.61SPCh. 23 - Prob. 23.62SPCh. 23 - Prob. 23.63SPCh. 23 - Prob. 23.64SPCh. 23 - Prob. 23.65SPCh. 23 - Draw the complete electron-dot structure and...Ch. 23 - Prob. 23.67SPCh. 23 - Prob. 23.68SPCh. 23 - Prob. 23.69SPCh. 23 - Prob. 23.70SPCh. 23 - Prob. 23.71SPCh. 23 - Prob. 23.72SPCh. 23 - Prob. 23.73SPCh. 23 - Prob. 23.74SPCh. 23 - Prob. 23.75SPCh. 23 - Prob. 23.76SPCh. 23 - Prob. 23.77SPCh. 23 - Prob. 23.78SPCh. 23 - Prob. 23.79SPCh. 23 - Prob. 23.80SPCh. 23 - Prob. 23.81SPCh. 23 - Prob. 23.82SPCh. 23 - Prob. 23.83SPCh. 23 - Prob. 23.84SPCh. 23 - Prob. 23.85SPCh. 23 - Prob. 23.86SPCh. 23 - What does it mean to say that fats and oils are...Ch. 23 - Prob. 23.88SPCh. 23 - Prob. 23.89SPCh. 23 - Prob. 23.90SPCh. 23 - Prob. 23.91SPCh. 23 - Prob. 23.92SPCh. 23 - Prob. 23.93SPCh. 23 - Prob. 23.94SPCh. 23 - Prob. 23.95SPCh. 23 - Prob. 23.96SPCh. 23 - Prob. 23.97SPCh. 23 - Prob. 23.98SPCh. 23 - Prob. 23.99SPCh. 23 - Prob. 23.100SPCh. 23 - Prob. 23.101SPCh. 23 - Prob. 23.102SPCh. 23 - Prob. 23.103SPCh. 23 - Prob. 23.104SPCh. 23 - Prob. 23.105SPCh. 23 - Prob. 23.106SPCh. 23 - Prob. 23.107SPCh. 23 - Prob. 23.108SPCh. 23 - Prob. 23.109SPCh. 23 - Prob. 23.110SPCh. 23 - Prob. 23.111SPCh. 23 - Prob. 23.112SPCh. 23 - Prob. 23.113SPCh. 23 - What amino acids do the following abbreviations...Ch. 23 - Name and draw the structures of amino acids that...Ch. 23 - Identify the amino acids present in the following...Ch. 23 - Prob. 23.117SPCh. 23 - Prob. 23.118SPCh. 23 - Prob. 23.119SPCh. 23 - Prob. 23.120SPCh. 23 - Prob. 23.121SPCh. 23 - Prob. 23.122SPCh. 23 - Prob. 23.123SPCh. 23 - Prob. 23.124SPCh. 23 - Prob. 23.125SPCh. 23 - Prob. 23.126SPCh. 23 - Prob. 23.127SPCh. 23 - Prob. 23.128SPCh. 23 - Prob. 23.129SPCh. 23 - Prob. 23.130SPCh. 23 - Prob. 23.131SPCh. 23 - Prob. 23.132SPCh. 23 - Prob. 23.133SPCh. 23 - Prob. 23.134MPCh. 23 - Prob. 23.135MPCh. 23 - Cytochrome c is an important enzyme found in the...Ch. 23 - Prob. 23.137MPCh. 23 - Prob. 23.138MP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- H-0-F: Draw the correct Lewis structure for HOF, including all lone pairs of electrons.arrow_forwardcan you state the number of electrons needed to reach octet? can you also state the difference between octet and valence electrons?arrow_forwarddiagram of a molecule that shows atoms based on their a periodic table symbols. These symbols are surrounded by pairs of dots to show valence electrons that could be lone pairs or bonding pairs?arrow_forward
- Write a Lewis structure for each of the following molecules. H3COCH3 (oxygen is between the two carbon atoms) Draw the molecule by placing atoms on the grid and connecting them with bonds. Include all lone pairs of electrons.arrow_forwardSF2 Draw the molecule by placing atoms on the grid and connecting them with bonds. Include all lone pairs of electrons and nonbonding electrons.arrow_forwardDraw the lewis dot diagram for iodine monochloride. How many total lone pairs of electrons are in this molecule?arrow_forward
- Write a Lewis structure for each of the following molecules. H2O2 Draw the molecule by placing atoms on the grid and connecting them with bonds. Include all lone pairs of electrons.arrow_forwardWrite a Lewis structure for each of the following molecules. H3COH (carbon and oxygen are both central) Draw the molecule by placing atoms on the grid and connecting them with bonds. Include all lone pairs of electrons.arrow_forwardIdentify the errors in each of the following Lewis structures and draw the correct formula. Draw the correct Lewis structure of this molecule by placing atoms on the canvas and connecting them with bonds. Include all hydrogen atoms and lone pairs of electrons.arrow_forward
- Write a Lewis structure for each of the following molecules. H2CO (carbon is central) Draw the molecule by placing atoms on the grid and connecting them with bonds. Include all lone pairs of electrons.arrow_forwardDraw 3 additional resonance structures that demonstrate which atoms have partial positive character.arrow_forwardH2CCCl2 Draw the molecule by placing atoms on the canvas and connecting them with bonds. Include all hydrogen atoms and nonbonding electrons.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY