
Concept explainers
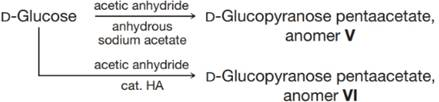
The
(a) Which proton in these anomers would be expected to have these highest
(b) Why do the signals for these protons appear as doublets?
(c) The relationship between the magnitude of the observed coupling constant and the dihedral angle (when measured using a Newman projection) between C−-H bonds on the adjacent carbons of a C−-C bond is given by the Karplus equation. It indicates that an axial–axial relationship results in a coupling constant of about 9 Hz (observed range is 8–14 Hz) and an equatorial–axial relationship results in a coupling constant of about 2 Hz (observed range is 1–7 Hz). Which of V and VI is the αa anomer and which is the
(d) Draw the most stable conformer for each of V and VI.
Want to see the full answer?
Check out a sample textbook solution
Chapter 22 Solutions
Organic Chemistry
Additional Science Textbook Solutions
CHEMISTRY-TEXT
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
Chemistry: A Molecular Approach (4th Edition)
Elementary Principles of Chemical Processes, Binder Ready Version
Living by Chemistry
Organic Chemistry As a Second Language: Second Semester Topics
- Draw the structure of C3H8O and indicate which sets of hydrogens correspond to which signal in the H NMR spectrum.arrow_forwardDraw the structure of C4H8O2 and indicate which sets of hydrogens correspond to which signal in the H NMR spectrum/table.arrow_forwardThe 1H NMR spectrum of CH3OH recorded on a 500 MHz NMR spectrometer consists of two signals, one due to the CH3 protons at 1715 Hz and one due to the OH proton at 1830 Hz, both measured downfield from TMS. (a) Calculate the chemical shift of each absorption. (b) Do the CH3 protons absorb upfield or downfield from the OH proton?arrow_forward
- When the 1HNMR spectrum of an alcohol is run in dimethylsulfoxide (DMSO) solvent rather than in chloroform, exchange of the Ο-H proton is slow and spin-spin splitting is seen between the Ο-H proton and C-H protons on the adjacent carbon. What spin multiplicities would you expect for the hydroxyl protons in the following alcohols? (a) 2-Methyl-2-propanol (b) Cyclohexanol (c) Ethanol (d) 2-Propanol (e) Cholesterol (f) 1-Methylcyclohexanolarrow_forwardThe carbonyl absorptions of esters X and Y differ by 25 cm−1. Whichcompound absorbs at higher wavenumber and why?arrow_forwardDetermine the correct structure of the compound, which shows following 13C NMR DEPT-135 data is 13C NMR DEPT-135: negative peaks at δ30.2, 31.9, 61.8, 114.7 ppm; positive peak at 130.4 ppm.arrow_forward
- Why is it better to acquire the 1H-NMR spectrum in DMSO-d6 and not in CDCl3?arrow_forwardInterpretation of H1 NMR of C6H12Oarrow_forwardThe 1H NMR spectrum of CH3OH recorded on a 500 MHz NMR spectrometer consists of two signals, one due to the CH3 protons at 1715 Hz and one due to the OH proton at 1830 Hz, both measured downeld from TMS. (a) Calculate the chemical shift of each absorption. (b) Do the CH3 protons absorb upeld or downeld from the OH proton?arrow_forward
- Identify the compound with molecular formulaC11H22O that gives the following 13C NMRspectrum:arrow_forwardgive structure of this c-nmr spectrumarrow_forwardThe 1H-NMR spectrum of compound B,C7H14O , consists of the following signals: δ 0.9 (t, 6H), 1.6 (sextet, 4H), and 2.4 (t, 4H). Draw the structural formula of compound B.arrow_forward
