
Organic Chemistry
12th Edition
ISBN: 9781118875766
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Question
Chapter 20, Problem 7PP
Interpretation Introduction
Interpretation:
Utilization of the reduction of an amide, oxime, or nitrile to carry out each of the given transformation, is to be shown.
Concept introduction:
▸ Aniline is a primary
The structure of aniline is shown as follows:
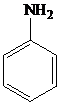
▸ Aniline and substituted aniline reacts with nitrous acid at low temperatures, to form benzene diazonium salts.
▸ The reduction of nitro group results in the formation of primary
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
(a) Explain why phentermine [PhCH2C(CH3)2NH2] can’t be made by a reductive amination reaction.(b) Give a systematic name for phentermine, one of the components of the banned diet drug fen–phen.
Show how to prepare the following aromatic amines by aromatic nitration, followed by reduction. You may use benzene andtoluene as your aromatic starting materials.(a) anilin
Show how to synthesize the following amines from the indicated starting materials byreductive amination.(a) benzylmethylamine from benzaldehyde
Chapter 20 Solutions
Organic Chemistry
Ch. 20 - Prob. 1PPCh. 20 - Prob. 2PPCh. 20 - Practice Problem 20.3
Write a mechanism that...Ch. 20 - Prob. 4PPCh. 20 - PRACTICE PROBLEM 20.5 Outline a preparation of...Ch. 20 - Prob. 6PPCh. 20 - Prob. 7PPCh. 20 - Prob. 8PPCh. 20 - Prob. 9PPCh. 20 - Prob. 10PP
Ch. 20 - Practice Problem 20.11 In the preceding examples...Ch. 20 - Prob. 12PPCh. 20 - Prob. 13PPCh. 20 - Practice Problem 20.14
Outline a synthesis of...Ch. 20 - Prob. 15PPCh. 20 - Prob. 16PPCh. 20 - Prob. 17PPCh. 20 - Prob. 18PPCh. 20 - Prob. 19PCh. 20 - 20.20 Give common or systematic names for each of...Ch. 20 - Which is the most basic nitrogen in each compound?...Ch. 20 - Prob. 22PCh. 20 - Prob. 23PCh. 20 - Show how you might synthesize each of the...Ch. 20 - Prob. 25PCh. 20 - 20.26 Provide the major organic product from each...Ch. 20 - Prob. 27PCh. 20 - 20.28 What products would you expect to be formed...Ch. 20 - Prob. 29PCh. 20 - Prob. 30PCh. 20 - Prob. 31PCh. 20 - Write equations for simple chemical rests or state...Ch. 20 - Prob. 33PCh. 20 - Explain the following, including mention of key...Ch. 20 - 20.35 Provide a detailed mechanism for each of the...Ch. 20 - Prob. 36PCh. 20 - Prob. 37PCh. 20 - Prob. 38PCh. 20 - Prob. 39PCh. 20 - 20.40 Give structures for compounds R-W:
Ch. 20 - Prob. 41PCh. 20 - Prob. 42PCh. 20 - Diethylpropion (shown here) is a compound used in...Ch. 20 - Prob. 44PCh. 20 - 20.45 Compound W is soluble in dilute aqueous HCI...Ch. 20 - 20.46 Propose structures for compounds X, Y, and...Ch. 20 - Compound A(C10H15N) is soluble in dilute HCI. The...Ch. 20 - Prob. 48PCh. 20 - Prob. 49PCh. 20 - For each of the following, identify the product...Ch. 20 - 20.51 Develop a synthesis for the following...Ch. 20 - 20.52 When phenyl isochiocyanatc, , is reduced...Ch. 20 - Prob. 53PCh. 20 - 20.54 Propose a mechanism that can explain the...Ch. 20 - When acetone is treated with anhydrous ammonia in...Ch. 20 - Prob. 56P
Knowledge Booster
Similar questions
- Show how to synthesize the following amines from the indicated starting materials.(a) N-cyclopentylaniline from anilinearrow_forwardShow how to prepare the following aromatic amines by aromatic nitration, followed by reduction. You may use benzene andtoluene as your aromatic starting materials. (c) m-bromoaniline (d) m-aminobenzoic acidarrow_forwardShow how you might utilize the reduction of an amide, oxime, or a nitrile to carry out each of the following transformations (a)Benzoic acid to N-ethyl-N-benzylamine (b)1-Bromopentane to hexylamine (c)Propanoic acid to tripropylamine (d)2-Butanone to sec-butylaminearrow_forward
- The two most general amine syntheses are the reductive amination of carbonyl compounds and the reduction of amides.Show how these techniques can be used to accomplish the following syntheses.(a) benzoic acid S benzylamine (b) benzaldehyde S benzylamine(c) pyrrolidine S N@ethylpyrrolidine (d) cyclohexanone S N@cyclohexylpyrrolidine(e) HOOC¬(CH2)3 ¬COOH S pentane@1,5@diamine (cadaverine)arrow_forwardArrange the members of each group in order of decreasing basicity: (a) Ammonia, aniline, methylamine (b) Acetanilide, aniline, N-methylaniline (c) 2,4-Dichloroaniline, 2,4-dimethylaniline, 2,4-dinitroaniline (d) 3,4-Dichloroaniline, 4-chloro-2-nitroaniline, 4-chloro-3-nitroaniline (e) Dimethylamine, diphenylamine, N-methylanilinearrow_forward(D)The best route to synthesise aromatic primary amines is by reduction of the corresponding nitro compounds. Draw the reaction scheme for the preparation of p-toluidine.arrow_forward
- Show how you would accomplish the following syntheses using amides as intermediates.You may use any necessary reagents.(a) benzoic acid S benzyldimethylaminearrow_forwardShow how to synthesize the following amines from the indicated starting materials byacylation–reduction. b) N-benzylaniline from anilinearrow_forwardChoose the correct phrase from the following One of the important methods for preparing cyclic amides is the reaction of Crignard reagent with cyclohexanone. The reaction of inulin with indole is given in an acidic medium that gives cyclic amide Caprolactam is a type of cyclic amide that can be prepared from cyclohexanearrow_forward
- Show how you would use appropriate acyl chlorides and amines to synthesize thefollowing amides.(a) N,N-dimethylacetamide (b) acetanilide (PhNHCOCH3)(c) cyclohexanecarboxamide (d)arrow_forwardShow how Gabriel syntheses are used to prepare the following amines.(a) benzylaminearrow_forwardAmobarbital is a sedative marketed under the trade name Amytal. Propose a synthesis of amobarbital, using diethyl malonate and urea as two of the starting materials.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
