
Interpretation:
Aniline is to be converted into the given desired product mentioned in each part.
Concept introduction:
Electrophiles are electron deficient species that have positive or partially positive charge. Lewis acids are electrophiles that accept electron pair.
Nucleophiles are electron rich species that have negative or partially negative charge. Lewis bases are nucleophiles that donate electron pair.
Free radical is an atom, molecule, or ion that has an unpaired electron, which makes it highly chemically reactive.
Substitution reaction: A reaction in which one of the hydrogen atoms of a hydrocarbon or a
Elimination reaction: A reaction in which two substituent groups are detached and a double bond is formed is called elimination reaction.
Addition reaction: It is the reaction in which unsaturated bonds are converted to saturated molecules by the addition of molecules.
Aniline is a primary
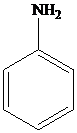
Aniline and substituted aniline reacts with nitrous acid at low temperature to form benzene diazonium salts.
The reduction of nitro group results in the formation of primary amine.
When aniline reacts with acetic anhydride, the product formed after the reaction is acetanilide.
When aniline reacts with Phthalimide
On nitration of acetanilide in the presence
When acetanilide reacts with chloro sulfuric acid, it forms p-acetamido benzene sulfonyl chloride.
Further, when p-acetamido benzene sulfonyl chloride reacts with ammonia, it gives rise to p-acetamido benzene sulfonamide, which on alkaline hydrolysis gives sulfanilamide.
When an aniline reacts with two moles of methyl iodide in the presence of a base, it forms N, N-dimethylaniline.
When an aniline reacts with nitrous acid at
When benzenediazonium halide reacts with copper(I) chloride, it forms chlorobenzene.
When benzenediazonium halide reacts with copper (I) bromide, it forms bromobenzene.
When benzenediazonium halide reacts with potassium iodide, it forms iodobenzene.
When benzenediazonium halide reacts with copper (I) cyanide, it forms benzonitrile.
Alkaline hydrolysis of benzonitrile results in the formation of benzoic acid.
When benzenediazonium halide reacts with copper oxide, it forms phenol.
When benzenediazonium halide reacts with Hypo phosphorous acid in the presence of moisture, it forms benzene.
Want to see the full answer?
Check out a sample textbook solution
Chapter 20 Solutions
Organic Chemistry
- Draw a structural formula for the product formed by treating butanal with each reagent. (a) LiA1H4LiA1H4 followed by H2OH2O (b) NaBH4NaBH4 in CH3OH/H2O (c) H2/Pt (d) Ag(NH3)2+in NH3/H2O (e) H2CrO4, heat (f) HOCH2CH2OH,HClarrow_forwardWhich is the stronger acid in each of the following pairs? Explain your reasoning. (a) Phenol or p-hydroxybenzaldehyde (b) m-Cyanophenol or p-cyanophenol (c) o-Fluorophenol or p-fluorophenolarrow_forwardShow how Gabriel syntheses are used to prepare the following amines.(a) benzylaminearrow_forward
- C) a nitrile D) an amine nitrite salt DIRECTIONS: Each of the following questions or incomplete statements below is followed by four suggested answers lettered A - D. Select or choose the letters of the best answer to the question or incomplete statement. 7. An organic nitrogen compound, X, gives ammonia on warming with dilute aqueous sodium hydroxide, X could be A) ethanamide B) ethylamine C) phenylamine D) amino ethanoic acid Which one of the following reagents reacts 1. in a similar fashion with both phenylamine (C H,NH,) and ethylamine. A) Br,(aq) C) conc. HNO, D) cold HNO (aq) 2. Phenylamine (aniline) can be prepared by reducing nitrobenzene with tin and concerntrated hydrochloric acid followed by the addition of alkali and finally steam distillation. The alkali is added to; A) prevent oxidation of phenylamine. B) liberate free phenylamine from solution C) dissolve excess nitro benzene D) dissolve the phenylamine 8. Arrange the following molecules in their increasing order of base…arrow_forward(a) How will you convert the following :(i) Propanone to Propan-2-ol (ii) Ethanal to 2-hydroxy propanoic acid(iii) Toluene to benzoic acid(b) Give simple chemical test to distinguish between :(i) Pentan-2-one and Pentan-3-one (ii) Ethanal and Propanalarrow_forwardSeveral additional amine syntheses are effectively limited to making primary amines. The reduction of azides and nitrocompounds and the Gabriel synthesis leave the carbon chain unchanged. Formation and reduction of a nitrile adds onecarbon atom. Show how these amine syntheses can be used for the following conversions.(a) allyl bromide S allylamine (b) ethylbenzene S p@ethylanilinearrow_forward
- The following three derivatives of succinimide are anticonvulsants that have found use in the treatment of epilepsy, particularly petit mal seizures. Q. Describe the chemistry involved in the conversion of (B) to (C). You need not present detailed mechanisms. Rather, state what is accomplished by treating (B) with NaOH and then with HCl followed by heating.arrow_forward1. Draw the major product of the reaction of 1-butanol and the following reagents. (a) PBr3 (b) SOCI2, py (c) HCI, ZNCI2 (d) Conc. H2SO4, heat (e) PCC, CH2CI2 (f) NażCr,O7, H2SO4, H2O (g) Li (h) NaH (i) TMSCI, Et3N (1) TSCI, pyridine (k) Na (1) Potassium tert-butoxidearrow_forwardVerapamil, a coronary artery vasodilator, is used in the treatment of angina caused by insufficient blood flow to cardiac muscle. Even though its effect on coronary vasculature tone was recognized over 30 years ago, only recently has its role as a calcium channel blocker become understood. Following is a retrosynthetic analysis leading to a convergent synthesis; it is convergent because (A) and (B) are made separately and then combined (i.e., the route converges) to give the final product. Convergent syntheses are generally much more efficient than those in which the skeleton is built up stepwise. Q. Two steps are required to convert (D) to (C). The first is treatment of (D) with ethyl chloroformate. What is the product of this first step? What reagent can be used to convert this product to (C)?arrow_forward
- Verapamil, a coronary artery vasodilator, is used in the treatment of angina caused by insufficient blood flow to cardiac muscle. Even though its effect on coronary vasculature tone was recognized over 30 years ago, only recently has its role as a calcium channel blocker become understood. Following is a retrosynthetic analysis leading to a convergent synthesis; it is convergent because (A) and (B) are made separately and then combined (i.e., the route converges) to give the final product. Convergent syntheses are generally much more efficient than those in which the skeleton is built up stepwise. Q. How do you account for the regioselectivity of the nucleophilic displacement involved in converting (C) to (B)?arrow_forwardThe saccharide shown here is present in some plant-derived foods. (a) Indicate the anomeric carbon atom(s) in this structure by drawing a circle around the atom(s) or by drawing an arrow pointing to the atom(s). (b) Would this saccharide give a positive result with Benedict’s reagent? Why or why not? (c) Would this saccharide give a positive result with Barfoed’s reagent? Why or why not? (d) Would this saccharide give a positive result with Seliwanoff’s reagent? Why or why not? (e) In a separate set of experiments, the saccharide solution was treated with a reagent that breaks glycosidic bonds. After this treatment, would any of the three assays give different results? Be sure to indicate which assay results would be different and give a reason.arrow_forwardRank the compounds in each of the following groups in order of their reactivity to electrophilic substitution: (a) Nitrobenzene, phenol, toluene, benzene (b) Phenol, benzene, chlorobenzene, benzoic acid (c) Benzene, bromobenzene, benzaldehyde, anilinearrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

