
Organic Chemistry
12th Edition
ISBN: 9781118875766
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 20, Problem 34P
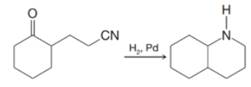
Explain the following, including mention of key intermediates that arc involved.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Observe and identify what's the general reaction scheme and explain it.
18.07a2
Add curved arrow(s) to draw the second step of the mechanism. Modify the given drawing of the products as needed to show the
intermediate that is formed in this step (a sigma complex). Use the +/- tools to add/remove charges, and use the single bond tool to
interconvert between double and single bonds.
The conversion of (CH3)3CI to (CH3)2C=CH2 can occur by either a onestepor a two-step mechanism, as shown in Equations [1] and [2].
Chapter 20 Solutions
Organic Chemistry
Ch. 20 - Prob. 1PPCh. 20 - Prob. 2PPCh. 20 - Practice Problem 20.3
Write a mechanism that...Ch. 20 - Prob. 4PPCh. 20 - PRACTICE PROBLEM 20.5 Outline a preparation of...Ch. 20 - Prob. 6PPCh. 20 - Prob. 7PPCh. 20 - Prob. 8PPCh. 20 - Prob. 9PPCh. 20 - Prob. 10PP
Ch. 20 - Practice Problem 20.11 In the preceding examples...Ch. 20 - Prob. 12PPCh. 20 - Prob. 13PPCh. 20 - Practice Problem 20.14
Outline a synthesis of...Ch. 20 - Prob. 15PPCh. 20 - Prob. 16PPCh. 20 - Prob. 17PPCh. 20 - Prob. 18PPCh. 20 - Prob. 19PCh. 20 - 20.20 Give common or systematic names for each of...Ch. 20 - Which is the most basic nitrogen in each compound?...Ch. 20 - Prob. 22PCh. 20 - Prob. 23PCh. 20 - Show how you might synthesize each of the...Ch. 20 - Prob. 25PCh. 20 - 20.26 Provide the major organic product from each...Ch. 20 - Prob. 27PCh. 20 - 20.28 What products would you expect to be formed...Ch. 20 - Prob. 29PCh. 20 - Prob. 30PCh. 20 - Prob. 31PCh. 20 - Write equations for simple chemical rests or state...Ch. 20 - Prob. 33PCh. 20 - Explain the following, including mention of key...Ch. 20 - 20.35 Provide a detailed mechanism for each of the...Ch. 20 - Prob. 36PCh. 20 - Prob. 37PCh. 20 - Prob. 38PCh. 20 - Prob. 39PCh. 20 - 20.40 Give structures for compounds R-W:
Ch. 20 - Prob. 41PCh. 20 - Prob. 42PCh. 20 - Diethylpropion (shown here) is a compound used in...Ch. 20 - Prob. 44PCh. 20 - 20.45 Compound W is soluble in dilute aqueous HCI...Ch. 20 - 20.46 Propose structures for compounds X, Y, and...Ch. 20 - Compound A(C10H15N) is soluble in dilute HCI. The...Ch. 20 - Prob. 48PCh. 20 - Prob. 49PCh. 20 - For each of the following, identify the product...Ch. 20 - 20.51 Develop a synthesis for the following...Ch. 20 - 20.52 When phenyl isochiocyanatc, , is reduced...Ch. 20 - Prob. 53PCh. 20 - 20.54 Propose a mechanism that can explain the...Ch. 20 - When acetone is treated with anhydrous ammonia in...Ch. 20 - Prob. 56P
Additional Science Textbook Solutions
Find more solutions based on key concepts
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
Consider a galvanic cell that uses the following half-reactions: 2H+(aq)+2eH2(g)Al3+(aq)+3eAl(s) a) What materi...
CHEMISTRY-TEXT
6.1 State the number of electrons that be must be lost by atoms of each of the following to achieve a stable el...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
This is to be explained how two different water vapor densities are at 100 % humidity. Concept Introduction : S...
Living by Chemistry
Q1. A chemist mixes sodium with water and witnesses a violent reaction between the metal and water. This is bes...
Chemistry: A Molecular Approach
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Kindly trace the arrow movement of its reaction mechanism thank youarrow_forwardWhat type of eliminaton reactions occur in this process?arrow_forward.. (a) Draw the mechanism for the hydrogenation reaction of olefins catalyzed by Schrock-Osborn complexes. Provide name of each and every step, also label the turnover-limiting step. catalytic [Rh(bisphosphine)] `Me H2 Me "Ме (b) Draw the mechanism for the hydrogenation of olefins catalyzed by Wilkinson's catalyst. Provide name of each and every step, also label the turnover-limiting step. catalytic Rh(PPH3)3CI Me H2 Me Mearrow_forward
- Experimental control is most clearly facilitated by means of…arrow_forwardGive the major organic product of the following reaction for letter e) and explain the mechanism/processarrow_forwardOzonolysis of one mole of the compound shown is expected to produce how many moles of CO, upon completion of the reaction?arrow_forward
- Draw the structure of the major organic product in the hydroboration‑oxidation reaction shown. (Sia = siamyl = sec‑isoamyl = 1,2‑dimethylpropyl.)arrow_forwardGive the synthesis pathway to synthesize the complex of trans-[PtCl2(en)(tu)] (structure below) by given the trans effect order is tu > Cl > NH3. H₂N H₂N tu= thiourea CI H₂N H₂N H₂arrow_forwardFor the reaction in part "b", please explain which products are the major and minor products. Also explain which are the kinetic and thermodynamic products.arrow_forward
- Outline the mechanism showing both products formed for the reaction of ammonia (2 mole equivalent) with 1-bromobutane (1 mole equivalent).arrow_forward9) A student described the following reaction as "I brominated it then added bulky base with heat". . A What would the different products be for the following different brominating reagents? NBS, Br₂, HBr, HBY/ROORarrow_forwardGive an example for each describe the following reactions (i) Hoffmann’s bromide reaction (ii) Gattermann reaction (in) A coupling reactionarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole
Seven Name Reactions in One - Palladium Catalysed Reaction (047 - 053); Author: Rasayan Academy - Jagriti Sharma;https://www.youtube.com/watch?v=5HEKTpDFkqI;License: Standard YouTube License, CC-BY