
Concept explainers
(a)
Interpretation:
Hybridization of underlined
Concept Introduction:
Hybridization is the hypothetical concept of mixing of atomic orbital into hybrid orbitals that are of dissimilar shapes, energies and are appropriate for combination of electrons to form bonds in
Hybridization is calculated by the hybrid orbitals and to calculate hybrid orbitals we need to know the steric number that is given by,
The table that relates the steric number with hybridization is as follows:
(a)
Answer to Problem 2F.8E
The molecule
Explanation of Solution
The molecule
The symbol for carbon is
The symbol for hydrogen is
Its Lewis structure is as follows:
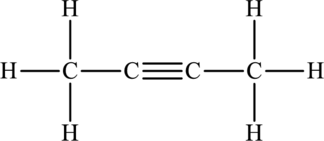
As from the structure above, 2 atoms are bonded to the underlined atom and 0 lone pair on it.
Substitute 2 for number of atoms bonded to the central atom and 0 for a number of lone pair on the central atom in equation (1) to find out the steric number.
Hence, the hybridization of
(b)
Interpretation:
Hybridization of underlined
Concept Introduction:
Refer to part (a).
(b)
Answer to Problem 2F.8E
The molecule
Explanation of Solution
The molecule
The symbol for carbon is
The symbol for hydrogen is
The symbol for nitrogen is
Its Lewis structure is as follows:
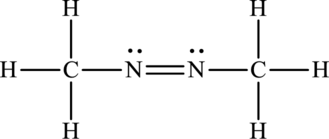
As from the structure above, 2 atoms are bonded to the underlined nitrogen atom and 1 lone pair on it.
Substitute 2 for a number of atoms bonded to the central atom and 1 for a number of lone pairs on the central atom in equation (1) to find out the steric number.
Hence, the hybridization of
(c)
Interpretation:
Hybridization of underlined
Concept Introduction:
Refer to part (a).
(c)
Answer to Problem 2F.8E
The molecule
Explanation of Solution
The molecule
The symbol for carbon is
The symbol for hydrogen is
Its Lewis structure is as follows:
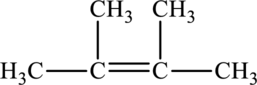
As from the structure above, 3 atoms are bonded to the underlined carbon atom and 0 lone pair on the same atom.
Substitute 3 for a number of atoms bonded to the central atom and 0 for a number of lone pair on the central atom in equation (1) to find out the steric number.
Hence, the hybridization of
(d)
Interpretation:
Hybridization of underlined
Concept Introduction:
Refer to part (a).
(d)
Answer to Problem 2F.8E
The molecule
Explanation of Solution
The molecule
The symbol for carbon is
The symbol for hydrogen is
The symbol for nitrogen is
Its Lewis structure is as follows:
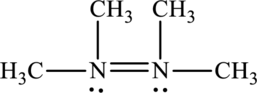
As from the structure above, 3 atoms are bonded to the underlined nitrogen atom and 1 lone pair on the same atom.
Substitute 3 for a number of atoms bonded to central atom and 1 for a number of lone pair on the central atom in equation (1) to find out the steric number.
Hence, the hybridization of
Want to see more full solutions like this?
Chapter 2 Solutions
Chemical Principles: The Quest for Insight
- Give the hybridization of each central atom in the following molecules. (a) cyclohexene (b) phosgene, Cl2CO (c) glycine, H2NC(1)H2C(2)OOH (Note: Numbers in parentheses label each carbon atom.)arrow_forwardIn each of the following molecules, a central atom is surrounded by a total of three atoms or unshared electron pairs: SnCl2, BCl3, SO2. In which of these molecules would you expect the bond angle to be less than 120? Explain your reasoning.arrow_forwardWhy do we hybtidize atomic orbitals to explain the bonding in covalent compounds? What type of bonds form from hybrid orbitals: sigma or pi? Explain.arrow_forward
- Specify the electron-pair and molecular geometry for each underlined atom in the following list. Describe the hybrid orbital set used by this atom in each molecule or ion. (a) CSe2 (b) SO2 (c) CH2O (d) NH4ssarrow_forwardSpecify the electron-pair and molecular geometry for each underlined atom in the following list. Describe the hybrid orbital set used by this atom in each molecule or ion. (a) BBr3 (b) CO2 (c) CH2Cl2 (d) CO32arrow_forwardWhy is the concept of hybridization required in valence bond theory?arrow_forward
- Consider the polyatomic ion IO65-. How many pairs of electrons are around the central iodine atom? What is its hybridization? Describe the geometry of the ion.arrow_forwardIdentify the orbitals on each of the atoms that form the bonds in H3CCN. How many bonds and bonds form?arrow_forwardWhat are the relationships among bond order, bond energy, and bond length? Which of these quantities can be measured?arrow_forward
- The structure of amphetamine, a stimulant, is shown below. (Replacing one H atom on the NH2, or amino, group with CH3 gives methamphetamine a particularly dangerous drug commonly known as speed.) (a) What are the hybrid orbitals used by the C atoms of the C6 ring. by the C atoms of the side chain, and by the N atom? (b) Give approximate values for the bond angles A, B, and C. (c) How many bonds and bonds are in the molerule? (d) Is the molecule polar or nonpolar? (e) Amphetamine reacts readily with a proton (H+) in aqueous solution. Where does this proton attach to the molecule? Explain how the electrostatic potential map predicts this site of protonation.arrow_forwardCalcium cyanamide, CaNCN, is used both to kill weeds and as a fertilizer. Give the Lewis structure of the NCN2 ion and the bonded-atom lone-pair arrangement and hybridization of the carbon atom.arrow_forwardtrans-tetrazene (N4H4) consists of a chain of four nitrogen atoms with each of the two end atoms bonded to two hydrogen atoms. Use the concepts of steric number and hybridization to predict the overall geometry of the molecule. Give the expected structure of cis-tetrazene.arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





