
Concept explainers
Interpretation:
Three possible structures for the constitutional isomers of molecular formula
Concept Introduction:
There are two main classes of isomers − constitutional isomers and stereo isomers.
Constitutional Isomers have the same molecular formula but differ in the way the atoms are connected to each other.
Chirality is the presence of an asymmetric carbon center in a molecule and a molecule which contains a chiral center cannot superimpose on its mirror image. To consider as chiral, molecule or object and its mirror image should not superimpose. To consider as achiral, molecule or object and its mirror image should be superimposed with each other.
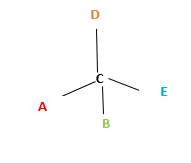
In the above diagram, where C is the chiral center/ asymmetric carbon center.
A, B, D, E are four different
Amine − is an organic N compounds formed by replacing one or more H atoms of
Want to see the full answer?
Check out a sample textbook solution
Chapter 18 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Draw the structure of a compound of molecular formula C4H11NO that ts each description: (a) a compound that contains a 1° amine and a 3° alcohol; (b) a compound that contains a 3° amine and a 1° alcohol.arrow_forwardThe following amines have the same molecular formula (C5H13N), but their boiling points are significantly different. Explain why. H `NH2 2-Methylbutan-1-amine Boiling point = 97 °C N-Methylbutan-2-amine Boiling point = 84 °C N-Ethyl-N-methylethan-1-amine Boiling point = 65 °Carrow_forward(a) Identify the functional groups in salinosporamide A, an anticanceragent isolated from marine sediment. (b) Classify each alcohol, alkylhalide, amide, and amine as 1°, 2°, or 3°.arrow_forward
- (a) Classify the carbon atoms in each compound as 1°, 2°, 3°, or 4°. (b) Classify the hydrogen atoms in each compound as 1°, 2°, or 3°.arrow_forward(a) Draw the structures for the eight constitutional isomers of molecular formula C 4H 11N. (b) Give the systematic name for each amine. (c) Identify the chirality center present in one of the amines.arrow_forwardBecause amines are basic, they can often accept a proton. Draw the protonated structure of n-propylamine.arrow_forward
- Vancomycin is a useful antibiotic for treating infections in cancer patients on chemotherapy and renal patients on dialysis. How many amide functional groups are present in vancomycin? Which OH groups are bonded to sp3 hybridized carbon atoms and which are bonded to sp2 hybridized carbons?arrow_forwardWhich compound(s) can hydrogen bond to another molecule like itself? Which compounds can hydrogen bond to water? (a) CH 3CONHCH 3; (b) HCON(CH 3) 2.arrow_forwardCycloserine is an antibiotic isolated from the microbe Streptomyces orchidaceous. It is used in conjunction with other drugs for the treatment of tuberculosis: S H-N NH₂ What is the molecular formula of this compound? Enter your answer in form of CxHyN₂Ow.arrow_forward
- What products are formed when benzoic acid (C 6H 5COOH) is treated with each base: (a) NaOH; (b) Na 2CO 3; (c) NaHCO 3?arrow_forward10 Jlgw What are the functional groups present in Aspirin which has the following structural formula aule ulao ut الدرجة من 1 C-OH lio ple P السؤال 0-C-CH3 alcohol , ketone and ether .a O carboxylic acid and ester .b O carboxylic acid , ether and ketone .c C aldehyde and ketone .dOarrow_forwardCompound X and compound Y are constitutional isomers with the molecular formula C5H10. Compound X possesses a carbon-carbon double bond in the trans configuration, while compound Y possesses a carbon-carbon double bond that is not stereoisomeric: Draw the structure of compound X. Draw Your Solutionarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
