
(a)
Interpretation:
The ammonium salt produced by the reaction between HCl and coniine alkaloid should be determined.
Concept Introduction:
Alkaloids are natural organic compounds present in plants. They are
Amines have basic properties, meaning they can act as proton acceptors.
Because of the basicity, amines react with acids such as HCl to form water-soluble ammonium salts. Below shown is the general reaction between a primary amine and HCl.
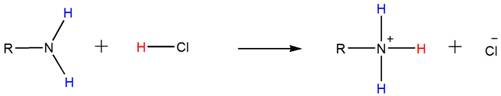
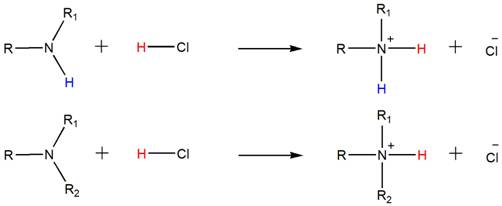
Accordingly, both the secondary and tertiary amines also form their respective ammonium salts with HCl.
(b)
Interpretation:
The ammonium salt produced by the reaction between HCl and morphine alkaloid should be determined.
Concept Introduction:
Alkaloids are natural organic compounds present in plants. They are amines.
Amines have basic properties, meaning they can act as proton acceptors.
Because of the basicity, amines react with acids such as HCl to form water-soluble ammonium salts. Below shown is the general reaction between a primary amine and HCl.
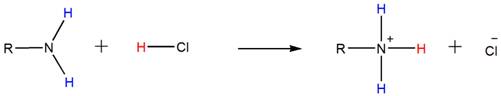
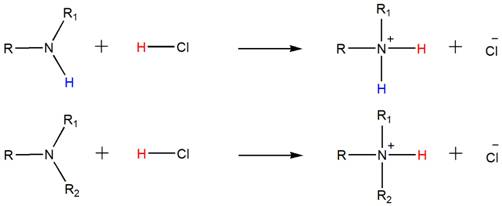
Accordingly, both the secondary and tertiary amines also form their respective ammonium salts with HCl.
Want to see the full answer?
Check out a sample textbook solution
Chapter 18 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Explin how you would separate a mixture containing benzoic acid and a neutral substance, benzophenone. Both compounds are insoluble in water and soluble in ether.arrow_forwardWhat is the role of phenolphthalein in the neutralization reaction? Draw the structure of phenolphthalein under acidic and basic conditions.arrow_forwardExplain why alkaloids do not have nomenclaturearrow_forward
- The other group are the alkaloids. Regardless of structure, alkaloids have a number of characteristics in common. What are TWO?arrow_forward1. What are the three kinds of alkaloids? Describe each. 2. Classify the following alkaloid according to ring system, draw the structure, then give the natural occurrence. a. betanidine b. peganine C. cocaine d. ergothioneinearrow_forwardThe following are central stimulants: Select one or more: A. Barbiturates. B. Morphine. C. Benzodiazepines. D. Cocaine. E. Amphetamines.arrow_forward
- In general, amines are nucleophilic due to: Select one: a. Existence of negative charge in amine group b. Amines are more electronegative than other functional groups C. Lone pairs in the amine group available for donation d. None of the abovearrow_forward3. Give the pharmacological property of the following alkaloids. Where do they naturally occur? a. morphine b. quinine c. berberine 4. What are the chemical tests used in identifying alkaloids?arrow_forwardWhat will happen to the body if the Ibuprofen and Aspirin will form an acid anhydrate inside it?arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,


