
Interpretation:
The way by which the given synthesis is being accomplished has to be shown and explanation for the stereoselective synthesis of 10-trans-12-cis isomer.
Concept introduction:
Wittig Reaction: It is an organic reaction where an
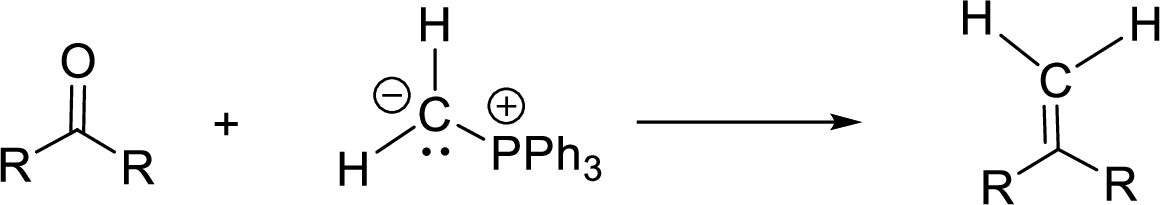
Non-stabilized ylide (having electron donating group on negatively charged carbon) gives Z-isomer whereas stabilized ylide (having electron withdrawing group on negatively charged carbon) gives E-isomer.
Non–stabilized ylide (having electron donating group on negatively charged carbon) gives Z–isomer whereas stabilized ylide (having electron withdrawing group on negatively charged carbon) gives E–isomer.
Trending nowThis is a popular solution!

Chapter 18 Solutions
Organic Chemistry
- Following is the structure of miconazole, the active antifungal agent in a number of over-the-counter preparations, including Monistat, that are used to treat vaginal yeast infections. One of the compounds needed for the synthesis of miconazole is the trichloro derivative of toluene shown on its right. (a) Show how this derivative can be synthesized from toluene. (b) How many stereoisomers are possible for miconazole?arrow_forwardThe following reaction was performed as part of a research program sponsored by the National Institutes of Health to develop therapeutic agents for the treatment of cocaine addiction. Using what you have seen about the reactions of halogens with alkenes, propose a mechanism for this process.arrow_forwardThe reaction of (S)-2-bromopentane with potassium cyanide to yield 2-methylpentanenitrile (2-cyanopentane) occurs via a nucleophilic substitution pathway. The reaction is 100% stereospecific.arrow_forward
- In a strongly acidic solution, cyclohexa-1,4-diene tautomerizes to cyclohexa-1,3-diene.Propose a mechanism for this rearrangement, and explain why it is energetically favorablearrow_forwardCompounds X and Y are both C7H15Cl products formed in the radical chlorination of 2,4-dimethylpentane. Base-promoted E2 elimination of X and Y gives, in each case, a single C7H₁4 alkene. Both X and Y undergo an SN2 reaction with sodium iodide in acetone solution to give C7H15l products; in this reaction Y reacts faster than X. What is the structure of X? • Do not use stereobonds in your answer. • In cases where there is more than one possible structure for each molecule, just give one for each. . Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate structures with + signs from the drop-down menu. наarrow_forwardThe electrophilic addition of HBr to 3-cyclohexylbut-1-ene gives a mixture of two constitutional isomers. These two isomers can also be produced from 3-cylcohexybut-2-ene, but one of which requires different reaction conditions; the electrophilic addition of HBr to 3-cylcohexybut-2-ene produces one of these isomers while the electrophilic addition of HBr to 3-cylcohexybut-2-ene, in the presence of peroxides, produces the other one. Draw the structure for the isomer product that can result from the reactants 3-cyclohexylbut-1-ene and 3-cylcohexybut-2-ene using different reaction conditions. Part 1 of 2 Click and drag to start drawing a structure. C 8 DY 000 8: >arrow_forward
- An aromatic hydrocarbon with a molecular formula of C13H20 has an 1 H NMR spectrum with a signal at ~7 ppm that integrates to 5H. It also has two singlets; one of the singlets has 1.5 times the area of the second. What is the structure of the aromatic hydrocarbon?arrow_forwardA CHEM 245 student attempts the following reaction to synthesize the desired ether below, but he is unsuccessful: H2SO4, CH3OH OCH3 desired ether racemic mixture (a) Use what you have learned in CHEM 241 to predict the structure of the major product he did collect: H2SO4, CH3OH (only one stereoisomer required) (b) Propose a reaction sequence that would be more likely to provide him with the desired ether product: OCH3 desired ether racemic mixture 3. Write out a complete mechanism for the reaction below: CH3 H3C CH3 H H Mechanism: 2.arrow_forwardCHEM 2252 Label the following compounds as aromatic, anti-aromatic, or non-aromatic (assume that all molecules are planar). In-class Assignment #4 Predict the product that would be formed when biphenyl reacts with a mixture of nitric and sulfuric acid. Draw a detailed, step-wise mechanism that shows formation of the electrophile and explains the observed regiochemistry. HNO3 H₂SO4arrow_forward
- A chemist requires a large amount of 1-bromo-5-methyl-2-hexene as starting material for a synthesis and decides to carry out the following NBS allylic bromination reaction in the presence of UV light. Draw the structures of all of the observed products.arrow_forwardProvide the reagent and mechanism for the transformation below. Explain in detail how the reaction conditions provide the given regioselectivity observed in the product.arrow_forwardIdentify products A and B from the given 1H NMR data. Treatment of acetone [(CH3)2C=O] with dilute aqueous base forms B. Compound B exhibits four singlets in its 1H NMR spectrum at 1.3 (6 H), 2.2 (3 H), 2.5 (2 H), and 3.8 (1H) ppm. What is the structure of B?arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

