
Concept explainers
(a)
Interpretation:
The structural formula for the principal product formed when benzamide is treated with the given reagent has to be drawn.
Concept introduction:
The normal reaction of an amide with water does not give good yield of product. As amides are least reactive
The reaction of water and amides in presence of acid gives a carboxylic acid as a product. The reaction equation is written as,
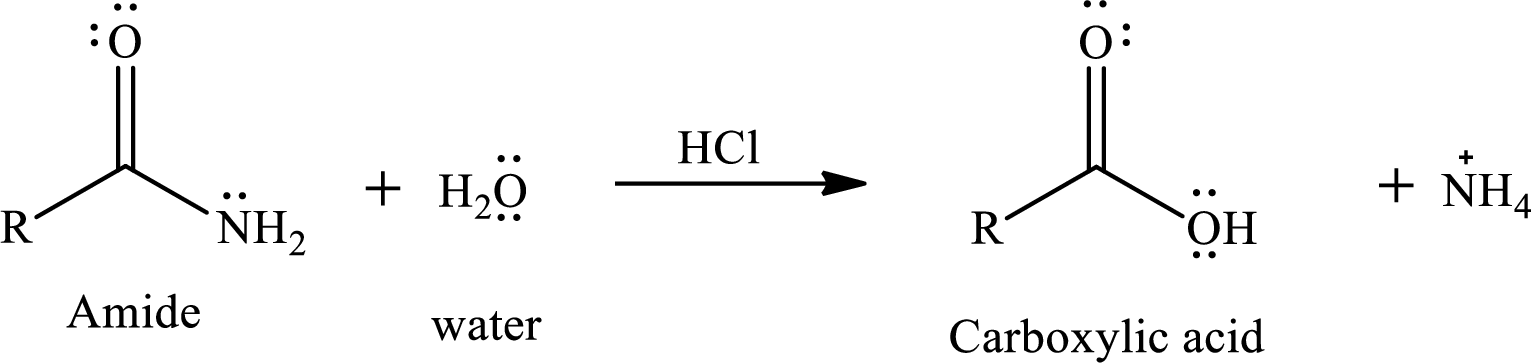
The reactivity of amide also depends upon the leaving tendency of amine part from acyl group. The weaker the base better will be the leaving group. The basicity of
(b)
Interpretation:
The structural formula for the principal product formed when benzamide is treated with the given reagent has to be drawn.
Concept introduction:
Amide hydrolysis in aqueous base:
A carboxylate salt and ammonia or an amine will be the product of amide hydrolysis in aqueous base.
For each mole of amide, one mole of base is required.
(c)
Interpretation:
The structural formula for the principal product formed when benzamide is treated with the given reagent has to be drawn.
Concept introduction:
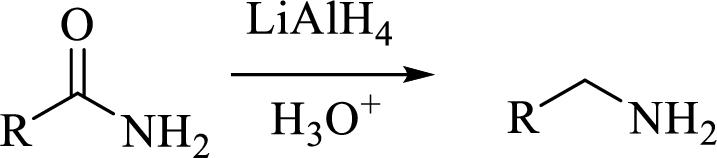
Trending nowThis is a popular solution!

Chapter 18 Solutions
Organic Chemistry
- Amines with more than 6 carbons are soluble in: a) aqueous HCI b) aqueous NaHCO3 d) water c) aqueous NaOH Which of the following would give a positive iodoform test? acetone a) benzophenone c) 3-pentanone d) cyclopentanone meth "Saponification" as the term is used in organic chemistry means: a) acidic hydrolysis of an ester b) basic hydrolysis of an ester c) acidic hydrolysis of an amide d) basic hydrolysis of an amide 3. ( Propylamine can be synthesized by the LiAlH4 reduction of: a) CH3CH2CECH b) CH3CH=NH d)) CH3CH2CEN c) CH3CH2NO2arrow_forwardIf you are given a mixture of three organic compounds (aspirin, β-naphthol, and naphthalene), what procedure would you follow to separate them?arrow_forwardDescribe concisely a chemical test to distinguish between the following pairs of compounds.(a) Propanal and propanone(b) Phenol and benzoic acid(c) Hexan-3-one and hexan-2-onearrow_forward
- Draw a structural formula for each amine. (a) 1,5-Pentanediamine (b) 2-Bromoaniline (c) Tributylaminearrow_forwardDraw the structures of the following compounds:(a) Ethanoic acid(b) Bromopentane(c) Butanonearrow_forwardDraw a structural formula for each amine. (a) 2-Butanamine (b) 1-Octanamine (c) 2,2-Dimethyl-1-propanamine (d) 1,5-Pentanediamine (e) 2-Bromoaniline (f) Tributylaminearrow_forward
- Draw compounds that contain the following: (a) A primary alcohol (c) A secondary thiol (c) An isopropyl group (b) A tertiary nitrile (d) A quaternary carbonarrow_forwardGive the chemical name, formula and use of the following esters: (a) Aspirin (b) Oil of Wintergreen (c) Salolarrow_forward(a) Compound Z is a tertiary aromatic amine with the formula, C8H11N. Provide a chemical structure for compound Z. (b)nDraw the structure of the product formed exclusively when nitrous acid reacts with Z.arrow_forward
- 1. Draw structures corresponding to the following IUPAC names: (a) 4-Methylpentanoic acid (b) o-Hydroxybenzoic acid (c) 2,2-Dimethylpropanoyl chloride (d) trans-2-Methylcyclohexanecarboxamide (e) p-Methylbenzoic anhydride (f) p-Bromobenzonitrilearrow_forwardDescribe concisely a chemical test to distinguish between the following pairs ofcompounds. (a) Pentan-2-one and pentan-3-one(b) Propanal and propanone(c) Phenol and anilinearrow_forwardHow would you use simple chemical tests to distinguish between the following pairs of compounds?(a) sodium stearate and p-dodecylbenzenesulfonate (b) beeswax and “paraffin wax”(c) trimyristin and myristic acidarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





