
Concept explainers
(a)
Interpretation:
The way by which the given conversion can be done in good yield has to be shown.
Concept introduction:
Acid chlorides are most often prepared by treating a
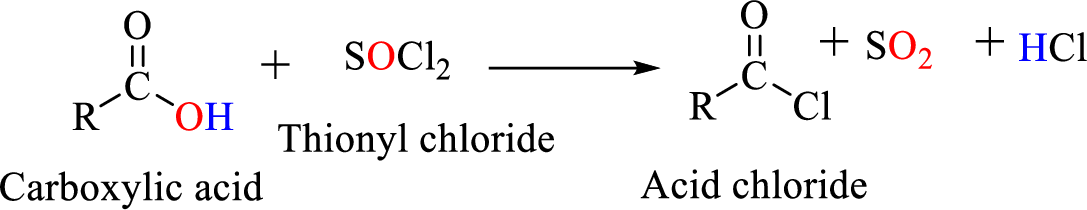
Gilman reagent:
It is generally represented as
General Reaction of Gilman reagent with carbonyl compounds:

(b)
Interpretation:
The way by which the given conversion can be done in good yield has to be shown.
Concept introduction:
Alkyl or aryl magnesium halides
Synthesis of Grignard reagent is shown below,
Addition of a Grignard reagent to carbon dioxide followed by protonation will produce carboxylic acid.
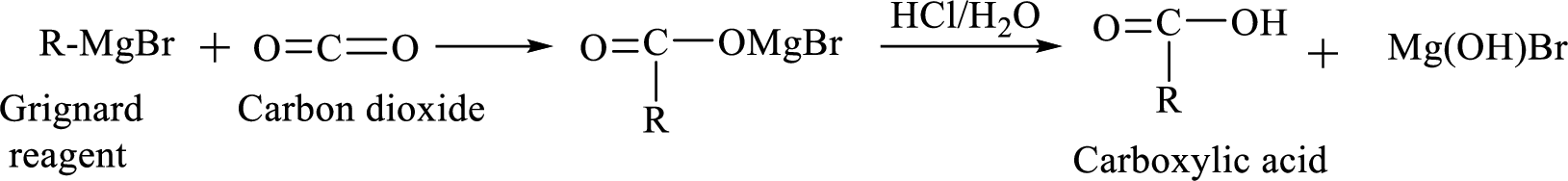
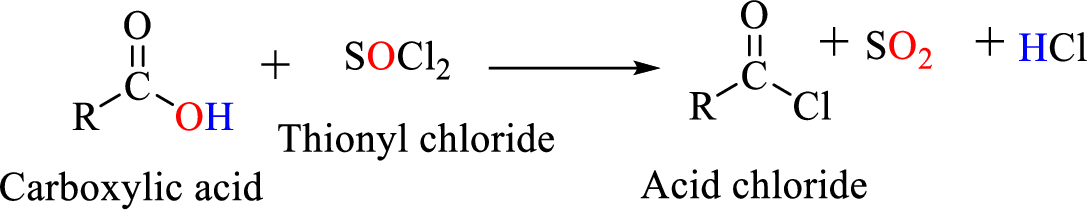
Acid chlorides are most often prepared by treating a carboxylic acid with thionyl chloride.
Want to see the full answer?
Check out a sample textbook solution
Chapter 18 Solutions
Organic Chemistry
- 3. Write all necessary chemical reactions. Name reactants & products. TOLLEN'S "SILVER MIRROR" TEST Acetaldehyde: Acetone: Formaldehyde: Methyl Ethyl Ketone (2-Butanone): Benzaldehyde: BENEDICT'S TEST Acetaldehyde: Acetone: Formaldehyde: Methyl Ethyl Ketone (2-Butanone): Benzaldehyde:arrow_forwardWhat reagents and reaction conditions are needed for each of the following conversions?arrow_forwardGiven the table , please calculate the theoretical and percentage yield of the starting material(cyclohexanol and sulfuric acid) and product(cyclohexene). densities: cyclohexanol-0.948 sulfuric acid-1.84 cyclohexene- 0.811arrow_forward
- 13)-Google Chrome od/quiz/attempt.php?attempt%315766088cmid%=813838&page312#question-1763030-41 - (Academic) Use one of the terms SNI, SN2, El or E2 to describe each of the following chemical reactions Choose the correct match for each NaNH, Choose... OTS NH3 CH;CH,O-Na+, Choose.. + ethanol КОН Но Br DMSO Choose...arrow_forwardClassify each reaction as addition, elimination, substitution. A) CH3CH2Br LIOH CH3CH2OH LİBR + B) CH3CH2CH2CI CH3CH=CH2 * HCI C) CH3CH=CH2 H2 CH;CH,CH3 H. H н—С— с—н + H-X + heat H. CI H H AB CD AD CB D)arrow_forwardPLEASE show steps for conversion and give written explanationarrow_forward
- 4. Show how you could make the following conversion. More than one step may be neededarrow_forwardCreate a hydrohalogenation reaction using a 5 carbon alkene illustrating Markovnikov's rule. Draw and name all reagentsarrow_forwardPd/C, H2 a. Propylcyclohexane O b. Propylbenzene OC. Benzoic acid O d.1-propenyl cyclohexanearrow_forward
- HgSO4 H2SO4, H20 1T (1) Br H2O2, NaOH 10 NaNH2 BH3, THE H =H 1S NH3 H20 HCI (1 equiv.) diethyl ether 1V 1W Br2 (m) MgBr (H,C=CH),CULI NaOCI ТСРВА > 1X 1Y > 1Z CH3COOH 1AA diethyl ether SoCI2, pyridine diethyl ether 1ABarrow_forwardConvert propan-2-ol [(CH3)2CHOH] to each compound. You may use any other organic or inorganic compounds.arrow_forwardFor each reaction, identify the starting material and reagent(s) that yield the product givenarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





