
(a)
Interpretation: Mechanism for the given type of decarboxylation reaction has to be proposed and it should be compared with the mechanism of decarboxylation of
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
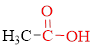
If
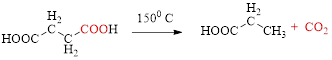
Decarboxylation of
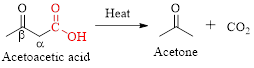
The mechanism of thermal decarboxylation involves two processes,
- (i) Redistribution of electrons in a cyclic transition state.
- (ii) Cyclic transition state possesses keto-enol tautomerism.
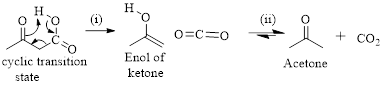
(b)
Interpretation: Mechanism for the given type of decarboxylation reaction has to be proposed and it should be compared with the mechanism of decarboxylation of
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,

If carboxylic acid is heated to a very high temperature then of carbon dioxide will be eliminated from it this reaction is known as decarboxylation. Simple carboxylic acids do not decarboxylate readily.
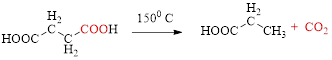
Decarboxylation of
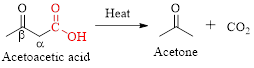
Simply the mechanism of thermal decarboxylation involves two processes,
- (i) Redistribution of electrons in a cyclic transition state.
- (ii) Cyclic transition state possesses keto-enol tautomerism.
Trending nowThis is a popular solution!

Chapter 17 Solutions
Organic Chemistry
- -Hydroxyketones and -hydroxyaldehydes are also oxidized by treatment with periodic acid. It is not the -hydroxyketone or aldehyde, however, that undergoes reaction with periodic acid, but the hydrate formed by addition of water to the carbonyl group of the -hydroxyketone or aldehyde. Write a mechanism for the oxidation of this -hydroxyaldehyde by HIO4.arrow_forwardFischer esterification cannot be used to prepare tert-butyl esters. Instead, carboxylic acids are treated with 2-methyIpropene in the presence of an acid catalyst to generate them. REFER IMAGE (a) Why does the Fischer esterification fail for the synthesis of tert-butyl esters?(b) Propose a mechanism for the 2-methylpropene method.arrow_forwardIn the presence of an appropriate base, esters undergo self-condensation to afford the corresponding B-ketoesters as shown below. Explain in detail using reaction schemes (not mechanisms) why route C and not routes A and B would lead to the expected product. 2 x Ethyl acetate A: NaOH(aq) B: OMe- C: OEt- EtO CH3arrow_forward
- 1. Br₂, PBrg 2. H₂O H₂C OH H3C OH Br The a-bromination of carbonyl compounds by Br₂ in acetic acid is limited to aldehydes and ketones because acids, esters, and amides don't enolize to a sufficient extent. Carboxylic acids, however, can be a-brominated by first converting the carboxylic acid to an acid bromide by treatment with PBr3. Following enolization of the acid bromide, Br₂ reacts in an α- substitution reaction. Hydrolysis of the acid bromide completes the reaction. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions H3C :0: :0::Br: Br Br H3C CO-P H Br Brarrow_forwardHemiacetals are formed by nucleophilic addition of a hydroxyl group to a carbonyl group. In the same way, cyclic hemiacetals can be formed by treating aldehyde with a catalytic amount of an acid. For the reaction below, draw the structure of the hemiacetal and write the possible mechanism for the transformation. H2SO4 но Harrow_forwardHow will you synthesize 6-hepten-2-one starting from ethyl 3 - oxobutanoate ? Write the sequence of the reactions involved and the required reagents.arrow_forward
- Write four examples of the condensation reactions of carbonyl compounds, starting with benzaldehyde (C6H5CHO), with appropriate reagents and appropriate reactions. Write down the mechanism of one you want in detail.arrow_forwardb) Clopirac has been used as a non-steroidal anti-inflammatory (NSAID) drug. The synthesis of clopirac involves reaction of 4-chloroaniline with hexan-2,5-dione in an initial step. Identify the named reaction that is taking place in this initial step and propose a mechanism for it. -NH₂ + $- Clopirac CO₂Harrow_forwardN Ph :0: CH3 Primary amines add to aldehydes and ketones to give imines. Imines are formed in a reversible, acid-catalyzed process that begins with nucleophilic addition of the primary amine to the carbonyl group, followed by transfer of the proton to yield a neutral carbinolamine. Protonation of the hydroxyl group converts it into a good leaving group and an E1-like loss of water yields an iminium ion. Deprotonation yields the product imine and regenerates the acid catalyst. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions CH3 NH₂OH NH₂OH Ph- :0: OH CH3 CH3 36arrow_forward
- 4) Show the mechanism for the acid catalyzed hydrolysis of the ketal shown below. کارام H+, H₂O HO OHarrow_forwardPhenyl 4-aminosalicylate is a drug used in the treatment of tuberculosis. Propose a synthetic route for this compound starting from 4-nitrosalicylic acid. The synthesis involves multiple steps. Specify all the reagents and the reaction conditions it may require in each step.arrow_forwardWrite the reagents to carry out the following reactions:arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
