
Concept explainers
(a)
Interpretation:
Show how to bring out the given conversion reaction in good yield.
Concept introduction:
Alkyl or aryl magnesium halides (RMgX) are known as Grignard reagent. The Grignard reaction is an organometallic
Synthesis of Grignard reagent is shown below,
The
Addition of a Grignard reagent to carbon dioxide followed by protonation will produce
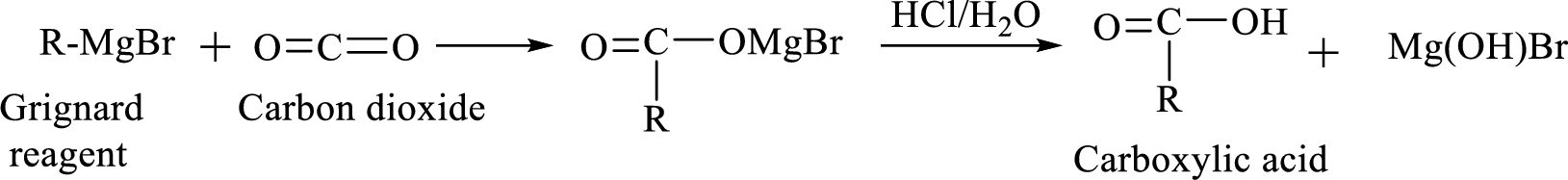
(b)
Interpretation:
Show how to bring out the given conversion reaction in good yield.
Concept introduction:
Carboxylic acid can be prepared from various ways; oxidation of
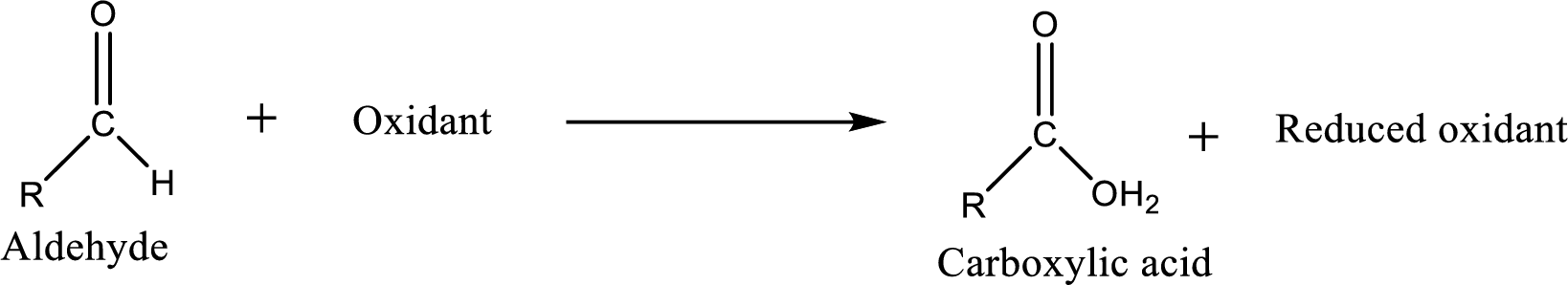
Carboxylic acid can be prepared from various ways; oxidation of aldehyde is one of the important methods to prepare carboxylic acid.
Carboxylic acid on further oxidation removes the carboxyl carbon as carbon dioxide. Depending on the reaction conditions, the oxidation state of the remaining organic structure may be higher, lower or unchanged.
Carboxylic acid can be prepared from primary alcohol by oxidation using strong oxidizing agents like chromic acid,
(c)
Interpretation:
Show how to bring out the given conversion reaction in good yield.
Concept introduction:
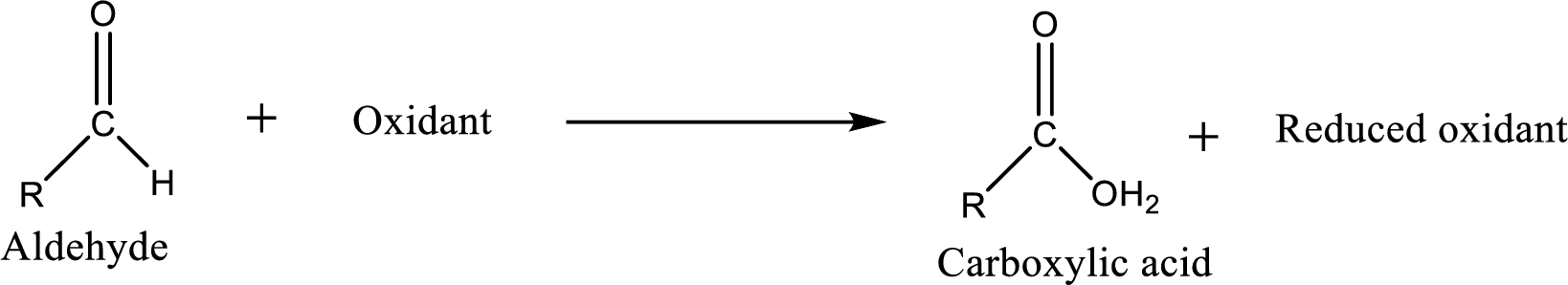
Carboxylic acid can be prepared from various ways; oxidation of aldehyde is one of the important methods to prepare carboxylic acid.
Carboxylic acid can be prepared from various ways; oxidation of aldehyde is one of the important methods to prepare carboxylic acid.
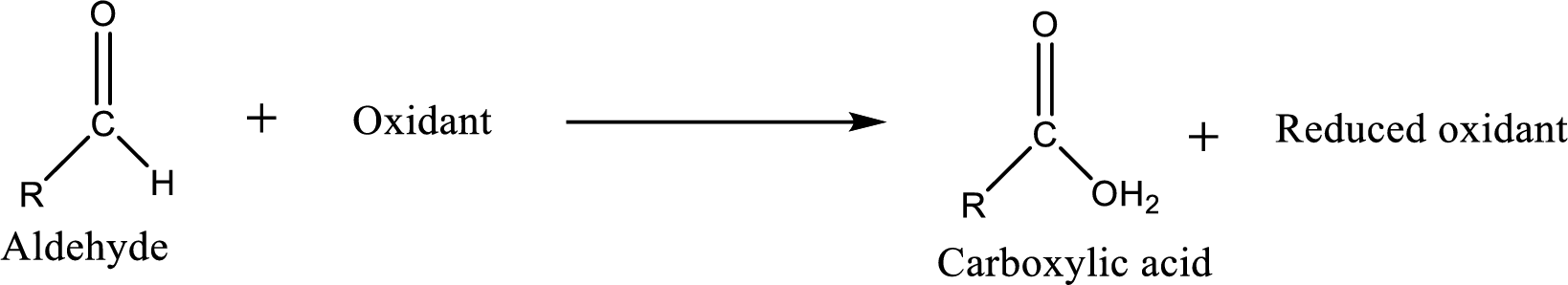
Carboxylic acid on further oxidation removes the carboxyl carbon as carbon dioxide. Depending on the reaction conditions, the oxidation state of the remaining organic structure may be higher, lower or unchanged.
Carboxylic acid can be prepared from primary alcohol by oxidation using strong oxidizing agents like chromic acid,
Trending nowThis is a popular solution!

Chapter 17 Solutions
Organic Chemistry
- 3. Write all necessary chemical reactions. Name reactants & products. TOLLEN'S "SILVER MIRROR" TEST Acetaldehyde: Acetone: Formaldehyde: Methyl Ethyl Ketone (2-Butanone): Benzaldehyde: BENEDICT'S TEST Acetaldehyde: Acetone: Formaldehyde: Methyl Ethyl Ketone (2-Butanone): Benzaldehyde:arrow_forwardGiven the table , please calculate the theoretical and percentage yield of the starting material(cyclohexanol and sulfuric acid) and product(cyclohexene). densities: cyclohexanol-0.948 sulfuric acid-1.84 cyclohexene- 0.811arrow_forward13)-Google Chrome od/quiz/attempt.php?attempt%315766088cmid%=813838&page312#question-1763030-41 - (Academic) Use one of the terms SNI, SN2, El or E2 to describe each of the following chemical reactions Choose the correct match for each NaNH, Choose... OTS NH3 CH;CH,O-Na+, Choose.. + ethanol КОН Но Br DMSO Choose...arrow_forward
- Classify each reaction as addition, elimination, substitution. A) CH3CH2Br LIOH CH3CH2OH LİBR + B) CH3CH2CH2CI CH3CH=CH2 * HCI C) CH3CH=CH2 H2 CH;CH,CH3 H. H н—С— с—н + H-X + heat H. CI H H AB CD AD CB D)arrow_forwardWhat reagents and reaction conditions are needed for each of the following conversions?arrow_forwardPLEASE show steps for conversion and give written explanationarrow_forward
- 4. Show how you could make the following conversion. More than one step may be neededarrow_forwardCreate a hydrohalogenation reaction using a 5 carbon alkene illustrating Markovnikov's rule. Draw and name all reagentsarrow_forwardPd/C, H2 a. Propylcyclohexane O b. Propylbenzene OC. Benzoic acid O d.1-propenyl cyclohexanearrow_forward
- Convert propan-2-ol [(CH3)2CHOH] to each compound. You may use any other organic or inorganic compounds.arrow_forwardHgSO4 H2SO4, H20 1T (1) Br H2O2, NaOH 10 NaNH2 BH3, THE H =H 1S NH3 H20 HCI (1 equiv.) diethyl ether 1V 1W Br2 (m) MgBr (H,C=CH),CULI NaOCI ТСРВА > 1X 1Y > 1Z CH3COOH 1AA diethyl ether SoCI2, pyridine diethyl ether 1ABarrow_forwardName the type of reaction and product of each reactions.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





