
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
8th Edition
ISBN: 9780134015187
Author: John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Question
Chapter 16, Problem 16.43AP
Interpretation Introduction
Interpretation:
It should be explained that why the given ammonium salt does not reacts with acid and base.
Concept introduction:
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
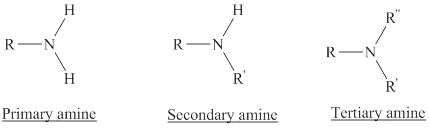
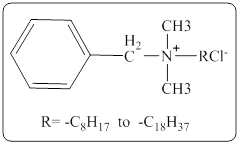
In a quaternary ammonium ion a nitrogen atom with four attached groups is positively charged. And their compounds are known as quaternary ammonium salt.
One commonly encountered quaternary ammonium salt has the following structure,
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
An amino acid mixture of phenylalanine, glycine and glutamic acid is to be separated by paper chromatography.The solvent is less polar than water. Which of these amino acids will have the highest Rf value and which the lowest? Explain.
An amino acid mixture of phenylalanine, glycine and glutamic acid is to be separated by paper chromatography. The solvent is less polar than water. Which of these amino acids will have the highest Rf value and which the lowest? Explain.
Cyanogen bromide is a common sulfhydryl-modifying reagent that can generally be used to break
disulfide bonds.
True
False
Chapter 16 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
Ch. 16.2 - Identify the following compounds as primary,...Ch. 16.2 - Prob. 16.2PCh. 16.2 - Prob. 16.3PCh. 16.2 - Prob. 16.4PCh. 16.2 - Prob. 16.5KCPCh. 16.2 - Prob. 16.6KCPCh. 16.3 - Arrange the following compounds in order of...Ch. 16.3 - Draw the structures of (a) ethylamine and (b)...Ch. 16.4 - Provide compounds that fit the following...Ch. 16.4 - Prob. 16.10P
Ch. 16.4 - Prob. 16.11PCh. 16.5 - Write an equation for the acid-base equilibrium...Ch. 16.5 - Prob. 16.13PCh. 16.5 - Prob. 16.14PCh. 16.5 - Prob. 16.15PCh. 16.5 - Prob. 16.16PCh. 16.6 - Prob. 16.17PCh. 16.6 - Prob. 16.18PCh. 16.6 - Prob. 16.19PCh. 16.6 - Prob. 16.20PCh. 16.6 - Prob. 16.21PCh. 16.6 - Prob. 16.22PCh. 16.7 - Prob. 16.1CIAPCh. 16.7 - Prob. 16.2CIAPCh. 16.7 - Prob. 16.3CIAPCh. 16 - (a) For the compound above, identify each nitrogen...Ch. 16 - The structure of the amino acid lysine (in its...Ch. 16 - Prob. 16.25UKCCh. 16 - Prob. 16.26UKCCh. 16 - Prob. 16.27UKCCh. 16 - Complete the following equations: (a) (b)...Ch. 16 - Prob. 16.29APCh. 16 - Draw the structures corresponding to the following...Ch. 16 - Name the following amines, and classify them as...Ch. 16 - Name the following amines, and identify them as...Ch. 16 - Prob. 16.33APCh. 16 - Which is a stronger base, diethyl ether or...Ch. 16 - Prob. 16.35APCh. 16 - Prob. 16.36APCh. 16 - The compound lidocaine is used medically as a...Ch. 16 - Prob. 16.38APCh. 16 - Draw the structures of the ammonium ions formed...Ch. 16 - Prob. 16.40APCh. 16 - Prob. 16.41APCh. 16 - Prob. 16.42APCh. 16 - Prob. 16.43APCh. 16 - Prob. 16.44APCh. 16 - Prob. 16.45CPCh. 16 - Prob. 16.46CPCh. 16 - Prob. 16.47CPCh. 16 - Prob. 16.48CPCh. 16 - How do amines differ from analogous alcohols in...Ch. 16 - Name at least two undesirable characteristics are...Ch. 16 - Prob. 16.52CPCh. 16 - Complete the following equations (Hint: Answers...Ch. 16 - Prob. 16.54CPCh. 16 - Prob. 16.55CPCh. 16 - Why is cyclohexylamine not considered to be a...Ch. 16 - Prob. 16.57CPCh. 16 - Prob. 16.58GPCh. 16 - 1-Propylamine, 1-propanol, acetic acid, and butane...Ch. 16 - Prob. 16.60GPCh. 16 - Lemon juice, which contains citric acid, is...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- Sodium laureth sulfate is found in many personal care products such as soap, shampoo, and toothpaste. Describe its function in these products and how it removes dirtarrow_forwardDiscuss why sodium hydroxide may be used in conjunction with triethanolamine to react with stearic acid in the formulation of vanishing creams.arrow_forwardAn amino acid mixture consisting of lysine, leucine, and glutamic acid is to be separated by ion-exchange chromatography, using a cationexchange resin at pH 3.5, with the eluting buffer at the same pH. Which of these amino acids will be eluted from the column first? Will any other treatment be needed to elute one of these amino acids from the column?arrow_forward
- Is sodium propanoate (the sodium salt of propanoic acid) as effective a soap as sodium stearate (the sodium salt of stearic acid)? Explain.arrow_forwardDoes it safe to use this sunscreen © on acne prone skin, and in peeling skin condition should we use sunscreen or not This sunscreen has lists of ingredients below (descending order)…. WATER CYCLOPENTASILOXANE ZINC OXIDE POLYGLYCERYL-3 POLYDIMETHYLSILOXYETHYL DIMETHICONE TRIETHOXYSILYLETHYL POLYDIMETHYLSILOXYETHYL HEXYL DIMETHICONE TITANIUM DIOXIDE SILICA / METHICONE ETHYLHEXYL METHOXYCINNAMATE PHENOXYETHYL CAPRYLATE BUTYLENE GLYCOL ZINC PCA GLYCERIN POLYGLYCERYL-10 LAURATE COCO-CAPRYLATE/CAPRATE DICTYOPTERIS MEMBRANACEA EXTRACT DICAPRYLYL CARBONATE LAURYL PEG-9 POLYDIMETHYLSILOXYETHYL DIMETHICONE DIETHYLAMINO HYDROXYBENZOYL HEXYL BENZOATE CAPRYLYL METHICONE POLYMETHYLSILSESQUIOXANE BIS-ETHYLHEXYLOXYPHENOL METHOXYPHENYL TRIAZINE Undecane (and) Tridecane PANTHENOL TOCOPHERYL ACETATE LENS ESCULENTA (LENTIL) SEED EXTRACT PEG-8 TOCOPHEROL ASCORBYL PALMITATE ASCORBIC ACID CITRIC ACID BISABOLOL PLANKTON EXTRACT DIMETHICONE CROSSPOLYMER CI 77492 TRIETHOXYCAPRYLYLSILANE STEARIC ACID CI 77491…arrow_forwardWhat is the purpose of: A. The concentrated salt solution? B. adding isopropyl alcohol to the solution? Why does it need to be in low temperature?arrow_forward
- What makes transparent softening lotion different from suspension softening lotion? Is it because of capric triglyceride makes it suspension?arrow_forwardWill ether test positive for translucent spot test of lipids? Why or why not?arrow_forwardGive the functions of the following ingredients, then name a branded/commercial skin or hair care product where the said material is used. Ozokerite Wax Sodium Ascorbate Lactic Acid 90% Titanium Dioxidearrow_forward
- Why is picric acid used for burns? Explain the principle involvedarrow_forwardBriefly explain why alpha-linolenic acid is essential.arrow_forwardIn a paragraph form, provide the experimental procedures of the development of the ring structure of α-tetralene by elimination of the water molecule will result in this product being heated in the presence of strong sulphuric acid.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Essentials of Pharmacology for Health ProfessionsNursingISBN:9781305441620Author:WOODROWPublisher:Cengage


Essentials of Pharmacology for Health Professions
Nursing
ISBN:9781305441620
Author:WOODROW
Publisher:Cengage


GCSE Chemistry - Acids and Bases #34; Author: Cognito;https://www.youtube.com/watch?v=vt8fB3MFzLk;License: Standard youtube license