
Concept explainers
Interpretation:
The structure of the given compound
Concept introduction:
In chemistry Structure is the arrangement of
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
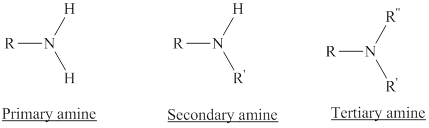
From the name of the compound its structure can be determined.
Primary amines can be named in the IUPAC system in several ways,
For simple amines the suffix – amine is added to the name of the alkyl substituent.
The suffix-amine can be used in place of the final –e in the name of the parent compound.
For a secondary amine an N prefixes the compound giving the shorter carbon chain and its chain prefix name.
For a tertiary amine an N, N prefixes the compound giving the two shorter carbon chains and their side chain prefix names.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- Methionine is an essential amino acid that can be metabolized to homocysteine. Using the figure below, describe the R-group on homocysteine and how it differs from methionine. Include the functionality of the thiol as part of this discussion.arrow_forwardSalol, the phenyl ester of salicylic acid, is used as an intestinal antiseptic. Draw the structure of phenyl salicylate.arrow_forwardDraw the structure of a triacylglycerol containing stearic acid, palmitic acid, and oleic acid.arrow_forward
- Salicylic acid (o-hydroxybenzoic acid) is used as starting material to prepare aspirin. Draw the structure of salicylic acid.arrow_forwardOxidation of the aldehyde group of ribose yields a carboxylic acid. Draw the structure of ribonic acid.arrow_forwardFigure 2 shows the structure of Diidopropylphosphofluoridate. Explain how this compound inhibits chymotrypsin. CH3 F CH3 H-C-0-P-0-C-H CH3 CH3 Diidopropylphosphofluoridate (DIPF) Figure 2arrow_forward
- Fumaric acid is a metabolic intermediate that has the systematic name trans-2-butenedioic acid. Draw its structure.arrow_forwardThe sugar alcohol often used in “sugarless” gums and candies is Lsorbitol. Much of this alcohol is prepared by reduction of Dglucose. Compare these two structures and explain how this can bearrow_forwardIn a paragraph form, provide the experimental procedures of the development of the ring structure of α-tetralene by elimination of the water molecule will result in this product being heated in the presence of strong sulphuric acid.arrow_forward
- Methionine has a sulfur atom in its formula. Explain why methionine does not form disulfide bridges.arrow_forwardDraw the structure of ethanolamine sphingomyelin formed from linoleic acid Draw the structure of serine sphingomyelin formed from arachidic acidarrow_forwardOlestra has been used in certain snack foods as an alternative to fats and oils. Its structure consists of a sucrose molecule in which all free hydroxyl groups have formed esters with oleic acid (an 18-carbon monounsaturated fatty acid). Olestra molecules have no caloric value because they are exceptionally large and cannot be digested. Draw the structure of olestra. Use R-COOH as an abbreviation for oleic acid.arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





